Concoction Preparation
Two commercially available fermented herbal concoctions were supplied by an herbalist (Mr. MS Mathebula) based at the University of Limpopo. Once procured, the fermented herbal concoctions were separately subjected to a stream of cold air to drive out any alcohol by products before freeze drying. The dried samples were partitioned into two from each herbal product, the crude extract while the other portion was extracted with 80% acetone. The crude extracts were reconstituted with water and the other with 80% aqueous ethanol. The former considered as the crude and the latter, regarded as a sub fraction. Table 1 list the plant material used to produce the herbal concoctions.
HIV-1 reverse transcriptase (RT) inhibitory bioassay
The effect of the herbal preparations on reverse transcription was evaluated using a non-radioactive HIV-RT colorimetric ELISA kit obtained from Roche Diagnostics, Germany and detailed by Ndhlala et al. (2010) with modifications (Matotoka et al., 2019). The protocol supplied together with the kit was followed, under nuclease-free conditions. The reverse transcriptase colorimetric assay takes advantage of the ability of RT to synthesize DNA, starting from the template/primer hybrid poly (A) × oligo (dT)15. The kit avoids the use of [3H]- or [32P]-labelled nucleotides which are used for the other classical RT assays. In place of radio-labelled nucleotides, digoxigenin- and biotin-labelled nucleotides are incorporated into one and the same DNA molecule, which is freshly synthesised by the RT. The detection and quantification of synthesized DNA as a parameter for RT activity is followed in a sandwich ELISA protocol: Biotin-labelled DNA freshly synthesised by the RT binds to the surface of microtiter plate modules (MPM) with wells that were precoated with streptavidin. In the next step, an antibody to digoxigenin, conjugated to peroxidase (anti-DIG-POD), binds to the digoxigenin-labelled DNA. In the final step, the peroxidase substrate ABTS (2, 2'-Azinobis [3-ethylbenzothiazoline-6-sulfonic acid]- diammonium salt) is added. The peroxidase enzyme catalyses the cleavage of the substrate, producing a coloured reaction product which is measured spectrophotometrically.
The following solutions provided with the kit were prepared according to the manufacturer; Solution 1, HIV-1 reverse transcriptase (final concentration 2 ng/μL, corresponding to 10 mU/μL) stored at -70°C. Solution 2, incubation buffer. Solution 3, reaction mixture containing poly (A) x oligo (dT)15 (46 mM Tris-HCl, 266 mM potassium chloride, 27.5 mM magnesium chloride, 9.2 mM DDT, 10 μM dUTP/dTTP, template/primer hybrid, 750 mA260 nm/mL). Solution 4, lysis buffer. Solution 5, anti-digoxigenin-peroxidase (anti-DIG-POD) (200 mU/mL). Solution 6, washing buffer and solution 7, ABTS substrate solution. In sterile Eppendorf tubes, 20 µL of resuspended herbal preparations (with final assay concentrations of 0.25, 2.5, 25, 250, 2500 µg/mL) or controls were mixed with 20 µL of recombinant HIV-1-RT (4 ng in lysis buffer) and 20 µL reaction mixture (solution 3) and the tubes were incubated for 1 hr at 37°C. After the 1 hr incubation period, the contents of the tubes (60 µL) were transferred into MPM wells. The MPM was covered with foil and incubated for 1 hr at 37°C after which the contents were removed from the MPM wells completely. The wells were rinsed 5 times with 250 μL of washing buffer (solution 6) per well for 30 s, with the washing buffer being removed carefully after each wash. After the wash, 200 μL of anti-DIG-POD (solution 5) was added to each well and the MPM was re-covered with foil and incubated for 1 h at 37°C. After the incubation period, the solution was removed completely from the MPM wells. The MPM wells were rinsed 5 times with 250 μL of washing buffer (solution 6) per well for 30 s, the washing buffer was removed carefully after each wash. After washing, 200 μL of ABTS substrate solution (solution 7) was added to each well and the MPM was incubated at room temperature for 5 minutes (a green colour appeared in the wells). The absorbance of the reaction mixture was then measured at 405 nm (reference wavelength: 490 nm) using a microplate reader (Opsys MRTM, Dynex Technologies Inc.). Percentage of inhibition was calculated by comparing the absorbance of the sample to the negative control using the equation below:
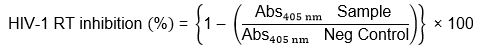
Where Abs405nm Sample is the absorbance of the reaction with herbal preparations or positive control at 405 nm and Abs405nm Neg Control is the absorbance of reaction with water instead of the sample at 405 nm.
Three tubes containing water instead of the sample were used as negative controls. Combivir® (GlaxoSmithKline) [lamivudine (1.0 mg/mL) + zidovudine (2.0 mg/mL)] and Kaletra® (Abbott) [lopinavir (8.9 mg/mL) + ritonavir (2.2 mg/mL)] were used as positive controls.
Results were presented as means duplicates ± standard deviations of 2 independent experiments; each experiment was done in duplicate. The IC50 values of herbal preparations were calculated using Graph Pad Prism (version 5.0).
Anti-inflammatory assay
Secretory phospholipase (sPLA2) inhibitory activity assay
The anti-inflammatory activity of the herbal concoctions was evaluated by examining the inhibition of the human sPLA2 enzyme using the sPLA2 (Type V) inhibitor screening assay kit (Cayman Chemical, Ann Arbor, MI) as described by George et al, (2014) with modifications (Thibane et al., 2019). The assay measures free 5-thio-2-nitrobenzoic acid (TNBA) following hydrolysis of diheptanoylthio-phosphatidylcholine (PC) by sPLA2. Briefly, 10 μL of the sPLA2 was dissolved in assay buffer solution (25 mM Tris–HCl, 10 mM CaCl2,100 mM KCl, 0.3 mM Triton X-100, pH 7.5) and 10 μL of concoction extract at concentrations of 50, 25, 12.5 and 6.25 μg/mL were added into a 96-well microtiter plate. The reaction was initiated by adding 200 μL of a substrate solution (diheptanoyl thio-PC, 1.66 mM), covered with aluminium foil and incubated at 25°C for 15 min. After incubation, 10 μL of DTNB (5,5′-dithio-bis-2-nitrobenzoic acid; 10 mM, 0.4 M Tris–HCl, pH 8.0) was added into each well. The reaction mixture containing the assay buffer and solvent served as the blank and mixture with sPLA2 enzyme and solvent served as the 100% initial activity (IA). Thioetheramide-PC served as the positive control. The hydrolysed diheptanoyl thio-PC was measured at 420 nm using a microplate reader (Optic Iveymen®System, Model 2100-C). The percentage inhibition was calculated using the formula below. The percentage inhibition was plotted against plant extract concentration and the IC50 was determined from the normalised logarithmic regression curve.
sPLA2 %Inhibition = [(100%IA−Inhibition) /100%IA ×100]
Where IA is inhibition absorbance
Lipoxygenase (15-LOX) inhibitory activity assay
The anti-inflammatory activity of the herbal concoctions was evaluated by examining the inhibition of the 15-LOX enzyme using the LOX inhibitor screening assay kit (Cayman Chemical, Ann Arbor, MI) as described by Boudjou et al, (2013) with modifications (Thibane et al., 2019). The assay measures hydroperoxides produced in the lipoxygenation reaction using purified 15-LOX. Briefly, 90 μL of 15-LOX was dissolved in assay buffer solution (0.1 M Tris–HCl, pH 7.4) and 10 μL concoctions extract at a concentration of (50, 25, 12.5 and 6.25 μg/mL) was added into each well of a 96-well microtiter plate. The plate was incubated at 25°C for 5 min. After incubation, the reaction was initiated by adding 10 μL substrate solution of arachidonic acid (1 mM) and mixed on a shaker for 10 min. The reaction was stopped by adding 100 μL of the chromogen into each well. The reaction mixture containing the assay buffer served as the blank and mixture with 15-LOX and solvent served as the 100% IA. Nordihydroguairetic acid (NDGA) served as a positive control. The absorbance was measured at 490 nm using a microplate reader (Optic Iveymen® System, Model 2100-C). The percentage inhibition was calculated using the formula below. The percentage inhibition was plotted against concoction extract concentration and the IC50 determined from the normalised logarithmic regression curve.
LOX % Inhibition = [(100% IA−Inhibition) /100%IA ×100]
Where IA is inhibition absorbance
Cyclooxygenase (COX-1 and COX-2) inhibitor screening assay
The anti-inflammatory activity of the two herbal concoctions was evaluated by examining the inhibition of the ovine COX-1 and human COX-2 enzyme using the COX inhibitor screening assay kit (Cayman Chemical, Ann Arbor, MI) as described by Boudjou et al, (2013) with modifications (Thibane et al., 2019). The assay measures the peroxidase activity of ovine COX-1 and human COX-2, by monitoring the appearance of oxidised N, N, N', N'-tetramethyl-p-phenylenediamine (TMPD). Briefly, 150 µL assay buffer (0.1 MTris-HCl, pH 8), 10 µL heme and 10 µL ovine COX-1 enzyme was added to each well of the 96-well microtiter plate. The same procedure was repeated with the human COX-2 enzyme. A volume of 10 µL concoction extract at a concentration of 50, 25, 12.5 and 6.25 µg/mL was added to each well. The plate was carefully mixed by shaking for 30 sec and followed by incubation at 25°C for 5 min. After incubation, 20 µL of TMPD was added to each well of the 96-well microtiter plate and the reaction was initiated by the addition of 20 µL arachidonic acid. The plates were further incubated at 25°C for 5 min. The reaction mixture containing assay buffer and heme served as the blank and a mixture with either COX-1 or COX-2 enzyme, assay buffer and heme served as the 100% IA. Indomethacin served as a positive control. The absorbance of oxidised TMPD was read at 490 nm using a microplate reader (Optic Iveymen® System, Model 2100-C). The percentage inhibition was calculated using the formula below. The percentage inhibition was plotted against concoction extract concentration and the IC50 was determined from the normalised logarithmic regression curve. Data were expressed as means of duplicates ± standard deviations.
COX % Inhibition = [(100% IA – Inhibitor)/100% IA x 100]
Where IA is inhibition absorbance.
Cytotoxicity and anti-cancer assay
To determine the toxicological outcomes of the consumption of the concoctions, their cytotoxic effect on normal human hepatoma cell lines (C3A). The anti-cancer effect of the herbal concoctions was evaluated on human colon (HT-29) cancer cells. The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) colorimetric assay described by Mosmann, (1983) was performed with modifications (Thibane et al., 2019). Human colon (HT-29) cancer cells and human hepatoma cell lines (C3A) (ATCC® HTB-38) (Biosafety level 2) were purchased from the American Type Culture Collection (ATCC). The cells were maintained in a flask with Dulbecco's Modified Eagle's Medium (DMEM), (Whitehead Scientific) supplemented with 10% v/v foetal bovine serum (FBS) (Adcock-Ingram) in a cell culture incubator at 37°C in humidified air containing 5% carbon dioxide (CO2). Cells (1×105 cells/mL) were seeded in a 96-well plate and allowed to attach overnight in a cell culture incubator. Cells were then treated with various concentrations (100-900 µg/mL) of different herbal concoctions for 24 hours. Following treatment, 20 µL of 5 mg/mL MTT was added and the cells were further incubated for 4 hours. The MTT solution was then removed and 200 µL of dimethyl sulfoxide (DMSO) was added to dissolve the MTT formazan crystals. Purple formazan crystals are formed when MTT is reduced by metabolically active cells. Thus, the number of formed formazan products produced indicates the number of viable cells. For quantification, the absorbance was measured at 560 nm using the GloMax®-Multi+Detection System microtitre plate reader (Promega).
Statistical analysis
The data were expressed as mean ± standard deviation (S.D). Statistically significant differences between the untreated control and treatments were determined using the GraphPad Software (Version 5, San Diego, CA). Samples were treated to one-way ANOVA, followed by Dunnett’s comparison test. Differences between means of untreated, treated cells were considered significant at p ≤ 0.05 (*) and highly significant at p ≤ 0.01 (**).