The current study demonstrated the feasibility of NACRT followed by MIE with an adequate R0 resection rate and acceptable local control as mid-term oncological outcomes for cT3br ESCC. Furthermore, patients who experienced postoperative complications had significantly lower RFS than those who did not. To the best of our knowledge, this is the first study to show that MIE after NACRT is feasible and that postoperative complications have a negative prognostic impact in patients with cT3br ESCC.
Surgery alone cannot control advanced esophageal cancer, and preoperative CF therapy was the standard treatment in Japan based on the findings of the JCOG9907 study [2]. Recently, the JCOG1109 study revealed a significant increase in survival in the preoperative docetaxel + cisplatin + 5-FU (DCF) therapy group compared to the preoperative CF group [3], and preoperative DCF therapy is now considered the new standard treatment for resectable ESCC in Japan [17].
However, when the tumor invades adjacent vital organs, definitive chemoradiotherapy is the main treatment option for T4b tumors [1], because combined resection of neighboring organs does not improve survival and has a high incidence of mortality and morbidity [24]. Some locally advanced tumors are difficult to diagnose as cT3 or cT4b and are commonly referred to as ‘borderline resectable ESCC’ [6–8]. The treatment strategy for these tumors has not been standardized, possibly due to the difficulty in determining whether it has adjacent organ invasion. Recently, attention has been drawn to the diagnosis of T classification in locally advanced ESCC [25], which revealed inter-observer variations in clinical diagnosis of the T category. The recently published Japanese Classification of Esophageal Cancer, 12th edition, categorized the clinical T category of locally advanced ESCC into cT3r, cT3br, and cT4b. The CT criteria for cT4b diagnosis were defined as, “flattening,” “deformity,” and “indentation” of the lumen of the trachea/bronchus, and “contact ≥ 110° for longer than 10 mm,” “contact of 90°–110° for longer than 20 mm,” or “encasement” for aortic invasion. We critically reviewed the pretreatment CT images using the diagnostic criteria described above [11–13], following the Japanese classification. The review was conducted independently and blindly by the 2 reviewers to avoid the study’s ambiguity.
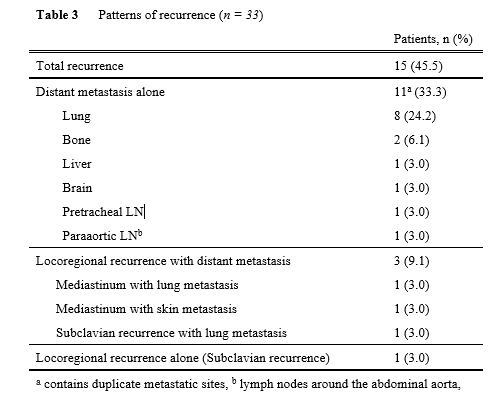
Although the standard treatment for cT3br tumors has not been established, the feasibility of NACRT followed by esophagectomy with the advantage of better local control for such tumors has been investigated [6–8]. The preferred chemotherapeutic regimen and the dose of radiotherapy were CF with 40Gy in general. R0 resection rates ranged from 44–85%, probably owing to variations in cT3br diagnosis. These studies used open esophagectomy, and there have been no studies using the MIE approach. After NACRT, the normal tissue gap disappears and is replaced by hard and fibrous tissue, particularly where the tumor invades the border of the preserving organs before treatment. Furthermore, strong adhesion to vital organs may result in lethal intraoperative complications. These factors may have prevented the use of MIE for this type of tumor. A magnified view in MIE has enabled more precise dissection, and in our study, there were no conversions, and we achieved a high R0 resection rate with similar postoperative complication rates as previous studies [7, 8]. Furthermore, locoregional recurrence alone was observed in only one case (3.0%), and our treatment strategy maintained acceptable locoregional control, even in this type of severely advanced tumor.
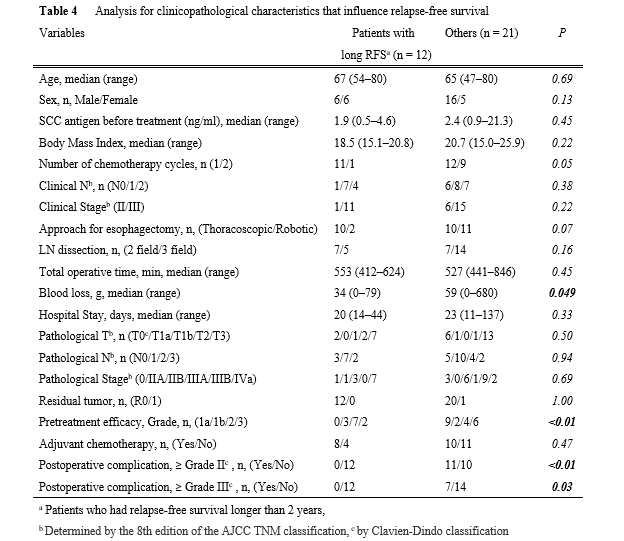
Despite good local control, 14 of the 15 recurrences had distant metastases. Good histological treatment efficacy could not predict survival, because 4 of 8 patients with Grade 3 pretreatment efficacy had distant metastases. Better pretreatment efficacy helped prevent relapse, as shown in Table 4, but sufficient survival could not be achieved even with complete local control. We had the possible explanations; this chemotherapeutic regimen lacked the power to control distant metastasis. To better control distant metastasis, other treatment strategies may be required. Recently, chemoselection of DCF and subsequent conversion surgery for clinical T4 tumors [26] provided favorable long-term outcomes with fewer distant metastases than ours. Strong chemotherapeutic regimens, such as DCF, may show promise for systemic control and better oncological outcomes.
Our research found that postoperative complications harmed survival. Similar to ours, previous research has suggested that postoperative complications worsened prognosis in patients with esophageal cancer [27–29]. Some studies found that pneumonia/infectious complications [27], anastomotic leakage [28], and all complications [29] had a negative prognostic impact. Changes in cytokines caused by postoperative complications may accelerate the development of microscopic residual disease into a clinically manifest recurrence. Postoperative complications would cause adjuvant chemotherapy to be delayed, and compliance would suffer as a result. In our study, the presence of whole postoperative complications (Grade ≥ II) worsened RFS. It will be interesting to see if the advantage of robotic surgery with refined visualization of surgical anatomy and precise movements of the stable articulated instruments, can be demonstrated in this patient cohort. In our study, recurrent laryngeal nerve palsy of Grade I was less common in RE than in TE (23% vs 55%, data not shown, p = 0.07). More research is needed to determine which clinicopathological factors are associated with postoperative complications, and it is critical to pursue reliable perioperative management to avoid complications.
There are several limitations to this study. First, this is a retrospective single-center study with a small number of patients, which may have resulted in selection bias. The number of preoperative chemotherapy cycles, MIE approach (thoracoscopic or robotic), and adjuvant chemotherapy indication and regimen have not been standardized. To reduce the selection bias in the inclusion of cT3br, the review process was conducted strictly. Second, the follow-up period was too short for a long-term analysis. Because the majority of relapses occurred within the first 2 years, patients with RFS ≥ 2 years were considered an “effectively treated population,” and were compared to other patients who may have had an insufficient follow-up period with the possibility of recurrence. We believe that our follow-up period was long enough to evaluate RFS. More research is needed to reveal solid long-term outcomes.
Finally, NACRT followed by MIE for cT3br tumors was feasible with sufficient R0 resection rates. In terms of mid-term oncological outcomes, it also provided adequate locoregional control but poor control of distant metastasis. Furthermore, postoperative complications harmed survival. NACRT followed by MIE for cT3br tumors would provide more acceptable oncological outcomes if we can successfully avoid postoperative complications.