T7 Expression System is a common method of ensuring tight control and high-level induced expression. However, this system can only work in some bacterial strains in which the T7 RNA Polymerase gene resides in the chromosome. In this study, we successfully introduced a chromosomal copy of the T7 RNA Polymerase gene under control of the lacUV5 promoter into Escherichia coli BW25113. The T7 Expression System worked efficiently in this mutant strain named BW25113-T7. We demonstrated that this mutant strain could satisfactorily produce 5-Aminolevulinic Acid via C5 pathway. A final study was designed to enhance the controllability of T7 Expression System in this mutant strain by constructing a T7 Promoter Variants Library. These efforts advanced E. coli BW25113-T7 to be a practical host for future metabolic engineering efforts.
Research
CRISPR/Cas9 mediated T7 RNA polymerase gene knock-in in E. coli BW25113 makes T7 expression system work efficiently
https://doi.org/10.21203/rs.3.rs-339217/v1
This work is licensed under a CC BY 4.0 License
Journal Publication
published 12 Aug, 2021
Read the published version in Journal of Biological Engineering →
You are reading this latest preprint version
E. coli
CRISPR/cas9
T7 Expression System
Fluorescent Protein
5-Aminolevulinic Acid
promoter variants
Metabolic engineering plays a critical part in bio-based production of fuels, chemicals and materials from biomass, and often involves integration of multiple genes to re-direct metabolic fluxes [1]. In recent years, biosynthesis techniques have attracted increased attention. As synthetic biology applications have grown in complexity, so too has the sophistication of available genetic and biochemical tools. For example, researchers started to produce antibiotic using microbial fermentation in the mid-20th century [2]. Currently, a dramatic change in the scope and complexity manufacturing processes within the space of synthetic biology has occurred. Thanks to development of genetic engineering, accomplishments have progressed from simple reconstitution of biosynthetic pathways in heterologous hosts to ambitious refactoring efforts [3–5] that can to produce high titers of medicinally relevant compounds (strictosamide, taxa-diene, etc.) [6, 7] or amino acids [L-valine, L-threonine, etc.] [8, 9]. Up to now, synthetic biologists have employed many tools to modify and reform bacterial strains to produce desired compounds. These tools include well-characterized sets of expression vectors [10], promoters for dynamic pathway control [11, 12], tunable protein degradation [13], and advanced methods for genome editing [14, 15].
Synthetic biologists prefer two organisms for expression and optimization of heterologous biosynthetic pathways: Escherichia coli and Saccharomyces cerevisiae. E. coli is the most widely used prokaryotic system that produces heterologous proteins for industrial production of bacterial metabolites by batch and fed-batch operations [16, 17]. E. coli is a gram-negative, facultative anaerobic and non-sporulating bacterium [18]. E. coli has been regarded as the workhorse of modern biotechnology [19] in the microbial production of biofuels and biochemicals.
To express heterologous proteins in E. coli, researchers have designed and developed a large number of expression vectors based on specific signals (such as promoters) that bacteria can recognize. One of the most frequently used expression systems is the T7 expression system. T7 is a kind of bacteriophage with highly efficient self-replication after infection of E. coli. Early transcription is initiated by T7 RNA polymerase which can strongly initiate transcription of other genes [20]. The T7 expression system has several advantages such as: T7 RNA polymerase can recognize the T7 promoter specifically and initiate transcription of downstream genes; the mRNA transcribed by the T7 expression system is stable and has a strong translational signal [20, 21].
To ensure the T7 expression system works in E. coli, researchers have integrated genes that code T7 RNA polymerase into the genome of some E. coli strains such as BL21(DE3) [20, 21]. However, there are still some E. coli strains which have special characteristics, but lack the T7 RNA polymerase gene. Consequently, it is of great significance to find a convenient tool to integrate a large gene (such as T7 RNA polymerase gene, more than 4 kb) into the bacterial genome. A possible solution is the CRISPR/Cas9 system which has been developed for programmable and customizable genome engineering [22].
CRISPR/Cas is an RNA-guided system which enables site-specific induction of a double strand break (DSB) and programmable genome editing. CRISPR/Cas is an immune system in bacteria and archaea [23]. Because of modularization and easy handling, CRISPR/Cas9 was adopted to engineer genomes of E. coli, lactic acid bacteria, streptomyces, Clostridium and so on [24]. Among CRISPR/Cas9 methods, recombineering based on λ-Red has been widely employed for genome manipulation [1], which necessitates an editing template donor DNA and three phage λ proteins: Exo, Bet and Gam [25]. Gam prevents the host RecBCD nuclease (a kind of exonucleases in bacteria) from degrading the linear donor DNA, while Exo exonuclease resects the double-stranded DNA (dsDNA) and Bet binds the single-stranded DNA (ssDNA) to promote annealing of ssDNA to homologous chromosomal sequences [26]. Although there are many reports of successful insertion, deletion and replacement of procaryotic genes using the CRISPR/Cas9 system, it is still difficult to integrate large pieces of DNA into an E. coli chromosome. In this study, we manage to integrate the T7 RNA polymerase gene into the chromosome of BW25113, which is a derivative of E. coli K-12 W1485. Subsequently, we test the efficiency of the T7 Expression System in this mutant strain to ensure that heterogeneous expression of protein occurs.
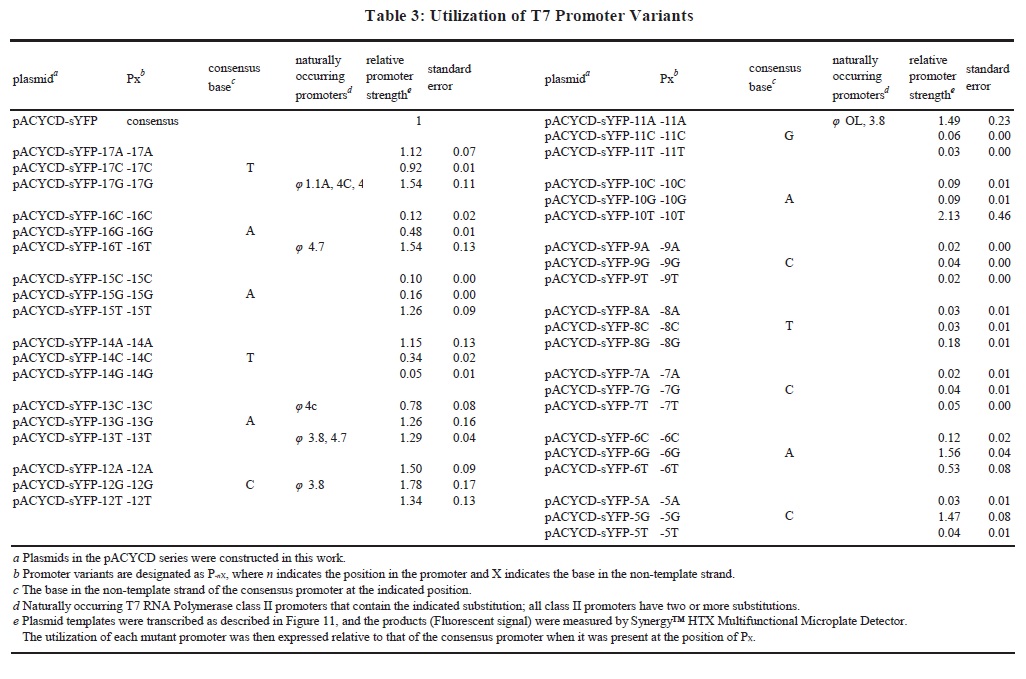
To make our findings more useful in metabolic engineering, we designed a library of T7-Lac promoter variants. Earlier studies indicated that the T7 promoter is composed of two functional domains: an upstream binding region from − 17 to -5 and an initiation region from − 4 to + 6. The promoter variants library was based on T7-Lac promoter [27], and the mutants were concentrated on the binding region (Fig. 1). We tested the expression strength of these promoter variants library in BW25113-T7, which will be useful in characterizing a number of aspects of promoter functions in metabolic engineering.
Selection of integration site and design of Left and Right Arm
T7 RNA polymerase gene, LacI gene, LacI Operator and Int 1 (T7-RNAP) were cloned from E. coli BL21(DE3). We inserted T7-RNAP into the ybhC gene site of BW25113 because we verified that the ybhC gene only encodes an outer membrane lipoprotein, which would not negatively effect in cell growth and metabolism after knocking out. The map of T7-RNAP in BL21(DE3) and the integration site in BW25113 are shown in Figs. 2A and 2B. The left homologous arm (HRL) was set to be the downstream of the ybhC gene (854–1255 bp), and the right homologous arm (HRR) was set in the middle of the ybhC gene (318–735 bp). Both homologous arms were approximately 400 bp, which could have a high efficiency of recombination [28]. The map of T7-RNAP integration in BW25113 in ideal condition is shown in Fig. 2C.
Construction of plasmids for CRISPR and preparation of linear Donor DNA
Plasmid pCas prepared by our lab harbored the temperature sensitive oriR101 with repA101ts for curing at 37℃, kanamycin resistance gene (Kanr), and the λ-Red operon encoding Gam, Bet, and Exo proteins under the control of arabinose-inducible promoter ParaB. This plasmid harbored S. pyogenes-derived cas9 driven by endogenous promoters and small-guide RNA (sgRNA) guided to ori-p15a which is under the control of lac operator (Fig. 3A).
Plasmid pTarget-ybhC was derived by targeting the pCRISPR crRNA region to the E. coli BW25113 ybhC gene, which harbored Chloramphenicol resistance (Chlr) and ori-p15a (Fig. 3B).
One difficulty was the acquiring the linear Donor DNA. The Donor DNA should contain the T7 RNA Polymerase gene and two homologous arms (HRL and HRR). The T7 RNA Polymerase gene contains about 4.5 kb making preparation of Donor DNA with high-fidelity difficult. In this research, we designed two intermediate cloning vectors for preparation of Donor DNA. We expected this approach would enhance efficiency of CRISPR/Cas9-mediated homologous recombination.
Primers used for preparation of Donor DNA are shown in Table 1. First, Fragment A (contained the ybhC gene, HRR, and HRL) was cloned from the E. coli BW25113 genome by Primer ybhC-F and ybhC-R. Then Fragment A was concatenated to pACYCD-Blank (p15a ori, Chlr) to assemble a new plasmid: pACYCD-ybhC (Fig. 4A and 4B). Next, T7-RNAP (Fragment B) was cloned from the E. coli BL21(DE3) genome by Primer RNAP-F and RNAP-R and then Fragment B was assembled with pACYCD-ybhC. Subsequently, linear Donor DNA could be cloned from this new plasmid named pACYCD-Donor (Fig. 4C and 4D). Donor DNA was purified, sequenced and confirmed to be without any mutations or absences.
Confirmation of CRISPR/Cas9-Mediated Gene Knock-in in BW25113
E. coli lacks the Non-Homologous End Joining (NHEJ) mechanism to survive DNA cleavage [29] so the most transformants harboring pCas and pTarget would die due to co-expression of Cas9, tracrRNA and crRNA, and subsequent DSB at the protospacer. Upon DNA damage, bacteria initiate a coordinated SOS response that involves activation of RecA protein and expression of error prone polymerase V encoded by umuDC to facilitate mutagenic DNA repair and cell survival [30]. Together with the editing template Donor DNA and λ-Red proteins, the SOS response can repair the DSB and the donor DNA could be mediated into the chromosome by homologous recombination [24, 25, 31].
To make CRISPR/Cas9-mediated homologous recombination in BW25113, we electroporated pCas (encoding both Cas and λ-Red proteins) into E. coli BW25113, followed by Arabinose (Ara) induction of pCas-encoded λ-Red proteins Gam, Bet and Exo. After preparing competent cells, pTarget-ybhC and Donor DNA were co-electroporated into cells (Fig. 5). The plate of cells after co-electroporating is shown in Figure S1.
To attest the integration into target locus, six bacterial colonies were selected for colony PCR (Figure S2). Primers for the test were Primer RNAP-R and Test-F (Table 1 and Table S1).
Subsequently, a rigorous examination was conducted to estimate whether T7-RNAP was integrated into the genome. The whole T7-RNAP fragment and adjacent chromosomal region (5496 bp; Fig. 2C) were cloned from this mutant strain by Primer Seq-F and Seq-R (Table S1). The PCR products were purified and sequenced to confirm that there were no mutations or absence in T7-RNAP. This mutant strain was named E. coli BW25113-T7.
Growth characteristic of E. coli BW25113-T7
Growth of E. coli BW25113-T7 in different medium was examined to assess whether CRISPR/Cas9-mediated T7-RNAP Knock-In affected the metabolic characteristics of the bacteria,
Growth of E. coli BW25113-T7 was compared with E. coli BW25113 and BL21(DE3). These three strains were cultured in standard M9 medium, special M9YE medium (standard M9 medium with additional 2 g/L Yeast Extract and 10 g/L Glucose) or standard LB medium (Fig. 6). There were no differences in growth rate among three strains in standard M9 medium (Fig. 6A). However, growth rate of E. coli BW25113 and BW25113-T7 were higher than strain BL21(DE3) in nutrient medium such as standard LB medium or special M9YE medium (Fig. 6B and 6C). Meanwhile, growth rate was similar between E. coli BW25113 and BW25113-T7 in these three types of medium (Fig. 6). These results indicated that induction of a chromosomal copy of the T7 RNA Polymerase gene does not impact growth characteristics of E. coli BW25113-T7.
Efficiency of T7 expression system in BW25113-T7
To explore whether the T7 expression system works in BW25113-T7, we detected fluorescence from the yellow fluorescent protein, sYFP, that is controlled by T7-Lac promoter (Fig. 7) in BW25113-T7 with IPTG induction (Fig. 8). The T7-Lac promoter did not activate expression of sYFP in E. coli BW25113 because it lacked T7 RNA Polymerase gene (Fig. 8A). After T7-RNAP knock-in, sYFP was expressed under control of T7-Lac promoter in E. coli BW25113-T7. Subsequently, the fluorescent signal could be macroscopically observed by Confocal Microscopy (Fig. 8B and 8C). This result indicated that this mutant strain did express heterologous proteins efficiently through the T7 Expression System.
he quantitative fluorescent signal was detected by a Multifunctional Microplate Detector (Fig. 9A). In E. coli BW25113-T7, the fluorescent signal was greater than that in E. coli BL21(DE3) (p < 0.001). Expression strength of sYFP under control of T7-Lac promoter in BW25113-T7 was about 4 times greater than BL21(DE3). These results revealed that the T7 expression system enhanced efficiency in E. coli BW25113-T7.
Production of 5-Aminolevulinic Acid via C5 pathway
To further ascertain the biosynthetic efficiency and metabolic engineering potential in E. coli BW25113-T7, we designed a process for 5-Aminolevulinic Acid biosynthesis in this mutant strain.
5-Aminolevulinic Acid (ALA), a five-carbon amino acid, is a key intermediate involved in the biosynthesis of tetrapyrrole [32]. ALA recently received much attention due to its potential applications in many fields, such as tumor-localization and photodynamic therapy for various cancers [33–35]. ALA is also used as a selective biodegradable herbicide and insecticide in agricultural applications due to its nontoxicity to crops, animals, and humans [36, 37]. Currently, biosynthesis of ALA has become the focus of much research [38]. In living organisms, there are two major pathways described for ALA biosynthesis: C4 pathway and C5 pathway [36]. The C5 pathway occurs in higher plants, algae, and many bacteria including E. coli [39]. In the C5 pathway, glutamate is the only substrate for biosynthesis of ALA. In E. coli, a metabolic strategy of ALA biosynthesis via C5 pathway has been developed [32]. Through this strategy, ALA can be synthesized in E. coli by over-expressing the key genes, hemA and hemL. In this research, we used a similar process for ALA biosynthesis (Fig. 10). In our mutant strain, the key genes, hemA and hemL, were activated efficiently and controlled by T7 Expression System.
Synthesis efficiency of ALA was compared among three strains of E. coli. After transferring pET28b-ALA-LA or pET28b-ALA-LAR (Fig. 10), cells were cultured in special M9YE medium. Genes, hemA and hemL, were over-expressed under control of T7-Lac promoter in expression vector pET28b-ALA-LA, while the exporter gene RhtA was over-expressed additionally in expression vector pET28b-ALA-LAR. After 24 h of fermentation, ALA concentration was determined (Fig. 9B). Concentration of ALA in the fermentation broth of BW25113-T7 was similar to that of BL21(DE3) when hemL and hemA were over-expressed. However, both of these two bacterial strains had a higher production of ALA when RhtA was else over-expressed. Notably, efficiency of ALA metabolism synthesis in BW25113-T7 was significantly higher than that of BL21(DE3) when these three key genes (hemA, hemL and RhtA) were over-expressed simultaneously by the T7 Expression System.
Construction of the T7 Promoter Variants Library
Plasmid pTarget-ybhC was constructed by inverse PCR and T4-Ligation (T4 DNA Ligase, NEB, England) to make the single-base substitution mutation (Fig. 7). The primers used are listed in Table S2. The plasmids were all sequenced to ensure that the single-base substitution mutation was introduced.
Data Analysis
Data for fluorescence intensity were subjected to analysis of variance (ANOVA) by GraphPad Prism (version 7.00). For fluorescent data, fluorescence intensity/OD600 was taken as the effective fluorescence intensity. Error bars indicate standard error of the mean (SEM). P values were calculated using Tukey's multiple comparisons test (*P < 0.05, **P < 0.01, ***P < 0.001).
As a common strategy of high-level induced expression, T7 expression system can ensure tight control to make the gene to be expressed in a plasmid under the control of a T7 phage promoter. T7 RNA polymerase is highly selective for its own promoters, which do not occur naturally in E. coli. A relatively small amount of T7 RNA polymerase provided from a cloned copy of T7 gene is sufficient to direct high-level transcription from a T7 promoter in a multicopy plasmid [20]. Specific mRNAs produced by T7 RNA polymerase are relatively stable and can rapidly saturate the translational machinery of E. coli, so that the rate of protein synthesis from the mRNA will depend primarily on efficiency of its translation. When mRNA is efficiently translated, a target protein can accumulate to greater than 50% of the total cell protein in three hours or less [20, 21].
To express heterologous proteins by the T7 Expression System, active T7 RNA polymerase was delivered to bacterial strains such as BL21(DE3) by induction of a chromosomal copy of T7 RNA Polymerase gene under control of the lacUV5 promoter (Fig. 2A). Expression plasmid vectors have been constructed to allow target genes to be placed under control of the T7-Lac promoter and to be expressed in bacteria strains which carry an inducible gene for T7 RNA Polymerase [27]. Transcription can be repressed strongly by lac repressor bound to an operator centered 15 base-pairs downstream from the RNA start. But, T7 RNA polymerase initiates transcription very actively from this T7-Lac promoter-operator combination in the absence of a repressor, or in the presence of a repressor plus inducer. To achieve a low basal expression of target genes, expression plasmid vectors usually carry a LacI gene that provides enough lac repressor to repress both the T7-Lac promoter in the multicopy vectors and the chromosomal gene for T7 RNA polymerase, which is controlled by the lacUV5 promoter. Upon induction, the usual high levels of expression are obtained [27, 40].
However, only strains with the T7 RNA Polymerase gene can use the T7 Expression System for efficient and controllable protein expression. Some strains commonly used in metabolic engineering (such as E. coli BW25113) cannot utilize the T7 Expression System even though they may have some superior characteristics (a higher growth rate or lower level of enterotoxin) [41, 42]. In this study, the CRISPR/Cas9 system combined with the λ-Red homologous recombination repair system was used to complete the insertion of the T7 RNA Polymerase gene into the genome of E. coli BW25113. We found that induction of a chromosomal copy of the T7 RNA Polymerase gene did not affect growth characteristics and enabled the T7 Expression System in this mutant strain for heterologous protein expression or gene over-expression.
The CRISPR/Cas9 system is an efficient and highly targeted gene editing tool. Since its discovery, Cas9 has been extensively used for genome editing in multiple organisms [43]. Although the full potential of CRISPR/Cas9 has not yet been harnessed, this technology has brought forth revolutionary changes in genomic research, including genome editing, regulation, and imaging [24, 44]. Despite Cas9’s great potential for both research and therapeutics, improvements can still be made in its specificity, efficiency, and spatiotemporal control [45]. There are still some challenges of CRISPR/Cas9 mediated large DNA Knock-In, such as high off-target rate, difficult access for Donor DNA, and high mutation rate of knock-in fragments [24, 46]. Based on the principle of ensuring fidelity of the repair template (Donor DNA), this study constructed two intermediate plasmids (pACYCD-ybhC and pACYCD-Donor) to acquire Donor DNA with high fidelity. The large DNA (T7-RNAP, 5.4kb) was inserted into the genome of E. coli BW25113, and this mutant strain was named BW25113-T7.
The working efficiency of the T7 expression system in E. coli BW25113-T7 was confirmed by Fluorescent-Protein-Based Reporter Gene System (Fig. 8). In this mutant strain, the T7 expression system expressed the sYFP fluorescent protein and the efficiency of protein expression was higher than that of BL21 (DE3) (Fig. 9A). Therefore, E. coli BW25113-T7 might provide the ability to produce high titers of simple peptides by the T7 Expression System.
The advantage of this protein expression could also extend to metabolic engineering. When key synthetase genes, hemA and hemL, were over-expressed by the T7 Expression System, there were no differences between BW25113-T7 and BL21 (DE3) in ALA production (Fig. 9B). This result is easily explained, as the accumulation of compounds is closely related to the activity and quality of synthetase in metabolic engineering [10, 32, 47]. However, when the exporter gene, RhtA, was over-expressed in addition to hemA and hemL, production of ALA in BW25113-T7 was significantly higher than that of BL21(DE3) (Fig. 9B). Therefore, we speculate that the T7 Expression System in E. coli BW25113-T7 has greater efficiency of expression efficiency than BL21(DE3). Rate of ALA export in the BW25113-T7 strain is higher than that of BL21 (DE3) due to greater expression of RhtA. As a result of accelerated ALA export, E. coli BW25113-T7 accumulated 42.4% more than E. coli BL21(DE3) (Fig. 9B).
Our mutant strain BW25113-T7 retains the characteristic rapid growth of the original strain. Although there were no differences of growth rate between BW25113-T7 and BL21(DE3) in minimal medium, BW25113-T7 showed faster growth when cultured in nutrient medium (Fig. 6). These results demonstrated that after T7-RNAP Knock-In, this mutant strain has certain advantages in metabolic engineering and synthetic biology. Otherwise, E. coli BW25113-T7 still has potential for genetic modification. This strain can be further edited and modified according to specific metabolic engineering strategies to make it more suitable for production of required compounds.
There are some disadvantages with the T7 Expression System. Due to the abnormally high activity of T7 polymerase, this inducible expression system is not the optimal choice for many applications in metabolic engineering. When the T7 Expression System was activated upon induction, a target protein can accumulate to greater than 50% of the total cell protein [20]. The high levels of protein expression would be a burden on cells, which leads to a negative impact on bacterial growth. Besides, synthetic biologists working in metabolic engineering are concentrated on activity and quality of synthetase not just quantity of synthetase [10, 32, 47]. Therefore, application of T7 Expression System still has some limitations in metabolic engineering. As an attempt to enhance controllability and practicability of the T7 Expression System in this mutant strain BW25113-T7 for metabolic engineering, we designed a series of experiments to construct a T7 Promoter Variants Library.
The essence of this T7 Promoter Variants Library is a series of promoter variants obtained by single-base mutation of each of the 13 bases on the Binding Region of the T7-Lac promoter (Fig. 1). Characterization of this series of promoters was measured by Fluorescent-Protein-Based Reporter Gene System (Fig. 11; Table 3). Based on such a library of promoters, expression of targeted proteins can be adjusted in a controlled manner such that application of the T7 expression system can be extended. We can flexibly regulate expression of targeted proteins according to specific needs to obtain the best yield of metabolites. For example, when we desired a higher intensity expression of heterologous protein, the T7-Lac promoter can be replaced as a variant with a higher expression intensity.
In conclusion, the T7 RNA Polymerase gene was inserted into E. coli BW25113 genome by CRISPR/Cas9 to construct a new E. coli strain in this study. This mutant strain maintained the advantages the original strain (such as the rapid growth rate) and allowed for efficient and controllable protein expression using the T7 Expression System. Further study of constructing T7 Promoter Variants Library enhanced the application of this strain in synthetic biology and provided a technical basis for industrial production in the future. Accordingly, these efforts advanced E. coli BW25113-T7 to be a practical host for future metabolic engineering efforts.
E. coli Strains
DH5α (TransGen, Beijing) was used for cloning plasmid. BL21(DE3) (TransGen, Beijing) was used for cloning T7 RNA polymerase genes, LacI and LacO (T7-RNAP), from its chromosomal genome. BW25113 strains were used for CRISPR/Cas9-induced DSB and recombination. Without especially explanations, all E. coli strains were routinely cultured in standard LB medium.
Selection Of Integration Site And Design Of Homologous Recombination
Sequence of T7-RNAP in BL21(DE3) genome and the integration site of BW25113 were both confirmed in NCBI. The function and detailed message of the ybhC gene was verified in NCBI and BioCyc Database. The N20 site was found by BROAD international design tool, which is available at: http://www.broadinstitute.org/rnai/public/analysis-tools/sgrna-design.
Plasmids Construction for CRISPR and preparation of linear Donor dsDNA by PCR
Table 1 contains all primer pairs we designed for gene cloning and intermediate plasmid construction. Plasmid pCas (Fig. 3A) and pTarget were prepared in our laboratory. Plasmid pTarget-ybhC was constructed by Inverse-PCR and T4-Ligation (T4 DNA Ligase, NEB, England) to replace the N20 fragment (Fig. 3B) with specific primers (Table 1). Plasmid pACYCD-ybhC and pACYCD-Donor were both constructed for preparing Donor DNA (Fig. 4) by In-Fusion® HD Cloning Kit (Takara, Japan). Donor DNA was cloned from pACYCD-Donor by Hi-Fi PCR (Phusion® High-Fidelity PCR Master Mix, NEB, England).
All plasmids used in this research will be listed in Table 2.
Electroporation, Cell Recovery, And Plating
For transformation, the plasmid or linear DNA were electroporated into competent cells in the pre-chilled cuvette (0.1 cm) using Bio-Rad MicroPulser (1.8 kV, time constant > 5.0 ms). For selection, 25 µg/mL chloramphenicol (Chl) or 50 µg/mL kanamycin (Kan) were used alone or in combination. For induction of λ-Red proteins and lac operator, 1 mM arabinose and 1 mM IPTG were used.
To prepare cells harboring pCas, cells cultured at 37℃ (OD600 = 0.45–0.55) were made competent, mixed with pCas (100 ng) and subjected to electroporation, after which the cells were recovered in SOC medium (1 mL) for 1 h at 30℃, plated onto the Kan plate, and cultured at 30 ℃ for 18–24 h.
For CRISPR/Cas9-mediated homologous recombination, cells harboring pCas were cultured at 30℃ in medium containing Kan and Arabinose and made competent. After co-electroporation of Donor DNA (400 ng) with pTarget-ybhC (100 ng), cells were recovered in SOC (1 mL) medium for 1 h at 30℃, plated onto Chl/Kan plate, and cultured at 30℃ for 18–24 h.
For elimination of pTarget-ybhC, cells harboring both pCas and pTarget were cultured at 30℃ in medium containing Kan and IPTG for 2 h. Cells were plated onto Kan plates and cultured at 30℃ for 18–24 h.
For elimination of pCas, cells harboring pCas were cultured at 37℃ in the medium without any antibiotic for 12–16 h. Then the cells were plated onto non-antibiotic plates and cultured at 37℃ for 12–16 h.
Verification Of Gene Integration By Colony PCR And DNA-SEQ
To verify correct integration of T7-RNAP, we designed Primer Seq-F and Primer Seq-R as shown in Table S1. Primer Seq-F targeted downstream of the ybhC gene (1267–1288 bp), while Primer Seq-R targeted upstream of the ybhC gene (261–282 bp). If integration was successful, colony PCR of the recombinants would yield a ≈ 5 kb amplicon, which contains HRL, T7-RNAP and HRR. Absences or mutations of the integration fragment were verified by DNA-sequencing.
All DNA-sequencing for this research was conducted by Beijing Ruibio BioTech Co., Ltd (China, Beijing).
Growth Of Bacteria In Different Medium
All E. coli strains were grown at 37℃ in conical flask (250 mL) containing M9 medium (100 mL), M9YE medium or LB medium with chloramphenicol (25 µg/mL). Growth was measured by monitoring optical density at 600 nm (OD600) using a spectrophotometer.
Confirmation of T7 Expression System in BW25113-T7 by sYFP Reporter Gene System
Efficiency of the T7 expression system was confirmed by a fluorescent protein reporter assay system. The plasmid was named as pACYCD-sYFP, which harbored ori-p15a, Chloramphenicol resistance (Chlr), LacI gene, T7-Lac promoter [27] and sYFP gene (Fig. 4). This fluorescent protein could reflect 540 nm light by the 503 nm exciting light. This optical signal could be observed microscopically by Confocal Microscopy and accurately detected by a Fluorescence Detector. This plasmid was transfected into E. coli BL21(DE3) (as Positive Control), E. coli BW25113 (as Negative Control) and E. coli BW25113-T7.
Confocal Microscopy
Cells harboring pACYCD-sYFP were cultured at 37℃ for 2 hours. Then IPTG with a final concentration of 1 mM was added to the induced group. The bacterium solution was observed under Confocal Microscopy after 4 hours of induction.
All strains were prepared for Confocal Microscopy as stated above for fluorescence measurements and prepared as a wet mount. Confocal microscopy was performed using a Leica TCS SP2 inverted confocal microscope (Leica Microsystems, Wetzlar, Germany) equipped with a 63× water-corrected objective in multitrack mode. Collected images were analyzed with the Leica Application Suite for Advanced Fluorescence (LAS AF, version 2.35) software (Leica Microsystem).
Fluorescence Intensity Measured By Multifunctional Microplate Detector
Cells harboring pACYCD-sYFP were cultured in LB containing Chl at 37℃ for 2 hours. Then IPTG with a final concentration of 1 mM was added to the induced group. The fermented liquid was added to a 96 well plate after 4 hours of induction, then accurate fluorescent data was detected by Synergy™ HTX Multifunctional Microplate Detector (BioTek Instruments, America). The excitation light was set as 503 nm and the receiving light was set as 540 nm. Collected data was analyzed with the Gen5™ V2 Data Analysis Software (BioTek Instruments, Inc.).
Analytical Procedures Of ALA Production
Cells harboring pET28b-ALA-LA or pET28b-ALA-LAR were cultured in M9YE containing Kan at 37℃ for 2 hours. Then IPTG with a final concentration of 0.1 mM was added to the induced group. To analyze ALA production, culture (30 mL) after induction for 24 h was centrifuged (12,000 g for 2 min at 4℃). The supernatant was used for extracellular ALA analysis. ALA concentration was analyzed using modified Ehrlich’s reagent [48].
E. coli: Escherichia coli; DSB: double strand break; dsDNA: double-stranded DNA; ssDNA: single-stranded DNA; T7-RNAP: T7 RNA polymerase gene, LacI gene LacI Operator and Int 1; HRL: left homologous arm; HRR: right homologous arm; Kanr: Kanamycin resistance gene; sgRNA: small-guide RNA; Chlr: Chloramphenicol resistance; NHEJ: Non-Homologous End Joining; ALA: 5-Aminolevulinic Acid
Acknowledgments
Not applicable.
Authors’ contributions
Qiao and Ye designed the study, wrote and edited the manuscript. Ye conducted all experiments of this study. Ye, Chen and Yang performed experiments and analyzed experimental results. Qiao and Zeng supervised the study. All authors read and approved the final manuscript.
Funding
The work presented in this manuscript was supported by Beijing Swine Innovation Team of Modern Agriculture Industry Technological System.
Data availability
The majority of data generated or analyzed during this study are included in this published article or in the supplementary information. The data not shown in the manuscript are available upon request from the corresponding author.
Ethics approval and consent to participate
Not applicable.
Consent for publication
The content of the manuscript has been approved by all authors.
Competing interests
The authors declare that they have no competing interests.
- Song CW, Lee J, Lee SY. Genome engineering and gene expression control for bacterial strain development. Biotechnol J. 2015;10:56-68.
- Baneyx F. Recombinant protein expression in Escherichia coli. Curr Opin Biotechnol. 1999;10:411-421.
- Yamanaka K, Reynolds KA, Kersten RD, Ryan KS, Gonzalez DJ, Nizet V, Dorrestein PC, Moore BS. Direct cloning and refactoring of a silent lipopeptide biosynthetic gene cluster yields the antibiotic taromycin A. Proc Natl Acad Sci U S A. 2014;111:1957-1962.
- Shao Z, Rao G, Li C, Abil Z, Luo Y, Zhao H. Refactoring the silent spectinabilin gene cluster using a plug‑and‑play scaffold. ACS Synth Biol. 2013;2:662-669.
- Temme K, Zhao D, Voigt CA. Refactoring the nitrogen fixation gene cluster from Klebsiella oxytoca. Proc Natl Acad Sci U S A. 2012;109:7085-7090.
- Brown S, Clastre M, Courdavault V, O’Connor SE. De novo production of the plant-derived alkaloid strictosidine in yeast. Proc Natl Acad Sci U S A. 2015;112:3205-3210.
- Soliman S, Tang Y. Natural and engineered production of taxadiene with taxadiene synthase. Biotechnol Bioeng. 2015;112:229-235.
- Park JH, Lee KH, Kim TY, Lee SY. Metabolic engineering of Escherichia coli for the production of L-valine based on transcriptome analysis and in silico gene knockout simulation. Proc Natl Acad Sci U S A. 2007;104:7797-7802.
- Lee KH, Park JH, Kim TY, Kim HU, Lee SY. Systems metabolic engineering of Escherichia coli for L-threonine production. Mol Syst Biol. 2007;3:149.
- Lee TS, Krupa RA, Zhang F, Hajimorad M, Holtz WJ, Prasad N, Lee SK, Keasling JD. BglBrick vectors and datasheets: A synthetic biology platform for gene expression. J Biol Eng 2011;5:12.
- Dahl RH, Zhang F, Alonso-Gutierrez J, Baidoo E, Batth TS, Redding-Johanson AM, Petzold CJ, Mukhopadhyay A, Lee TS, Adams PD, Keasling JD. Engineering dynamic pathway regulation using stress-response promoters. Nat Biotechnol. 2013;31:1039-1046.
- Zhang F, Carothers JM, Keasling JD, Joint Bioenergy I. Design of a dynamic sensor-regulator system for production of chemicals and fuels derived from fatty acids. Nat Biotechnol. 2012;30:354-359.
- Cameron DE, Collins JJ. Tunable protein degradation in bacteria. Nat Biotechnol. 2014;32:1276-1281.
- Wang HH, Isaacs FJ, Carr PA, Sun ZZ, Xu G, Forest CR, Church GM. Programming cells by multiplex genome engineering and accelerated evolution. Nature. 2009;460:894-898.
- Hsu PD, Lander ES, Zhang F. Development and applications of CRISPR-Cas9 for genome engineering. Cell. 2014;157:1262-1278.
- Lee SY. High cell-density culture of Escherichia coli. Trends Biotechnol. 1996;14:98-105.
- Yee L, Blanch HW. Recombinant protein expression in high cell density fed-batch cultures of Escherichia coli. Biotechnology (N Y) . 1992;10:1550-6.
- Singleton P. Bacteria in biology, biotechnology and medicine. 1999;5 ed, vol 157. Wiley.
- Himmel ME, Karplus PA, Sakon J, Adney WS, Baker JO, Thomas SR. Polysaccharide hydrolase folds diversity of structure and convergence of function. Appl Biochem Biotechnol. 1997;63-65:315-325.
- Studier FW, Moffatt BA. Use of bacteriophage T7 RNA polymerase to direct selective high-level expression of cloned genes. J Mol Biol. 1986;189:113-130.
- Studier FW, Rosenberg AH, Dunn JJ, Dubendorff JW. Use of T7 RNA polymerase to direct expression of cloned genes. Methods Enzymol. 1990;185:60-89.
- Jinek M, Chylinski K, Fonfara I, Hauer M, Doudna JA, Charpentier E. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science. 2012;337:816-821.
- Horvath P, Barrangou R. CRISPR/Cas, the immune system of bacteria and archaea. Science. 2010;327:167-170.
- Chung ME, Yeh IH, Sung LY, Wu MY, Chao YP, Ng IS, Hu YC. Enhanced integration of large DNA into E. coli chromosome by CRISPR/Cas9. Biotechnol and bioeng. 2017;114:172-183.
- Yu D, Ellis HM, Lee EC, Jenkins NA, Copeland NG, Court DL. An efficient recombination system for chromosome engineering in Escherichia coli. Proc Natl Acad Sci U S A. 2000;97:5978-5983.
- Court DL, Sawitzke JA, Thomason LC. Genetic engineering using homologous recombination. Annu Rev Genet. 2002;36:361-388.
- Dubendorff JW, Studier FW. Controlling basal expression in an inducible T7 expression system by blocking the target T7 promoter with lac repressor. J Mol Biol. 1991;219:45-59.
- Pyne ME, Moo-Young M, Chung DA, Chou CP. Coupling the CRISPR/Cas9 system with lambda red recombineering enables simplified chromosomal gene replacement in Escherichia coli. Appl Environ Microbiol. 2015;81:5103-5114.
- Wilson TE, Topper LM, Palmbos PL. Non-homologous end-joining: bacteria join the chromosome breakdance. Trends Biochem Sci. 2003;28:62-66.
- Michel B. After 30 years of study, the bacterial SOS response still surprises us. PLoS Biol. 2005;3:e255.
- Cong L, Ran FA, Cox D, Lin S, Barretto R, Habib N, Hsu PD, Wu X, Jiang W, Marraffini LA, Zhang F. Multiplex genome engineering using CRISPR/Cas systems. Science. 2013;339:819-823.
- Kang Z, Wang Y, Gu P, Wang Q, Qi Q. Engineering Escherichia coli for efficient production of 5-aminolevulinic acid from glucose. Metab Eng. 2011;13:492-498.
- Bhowmick R, Girotti AW. Cytoprotective induction of nitric oxide synthase in a cellular model of 5-aminolevulinic acid-based photodynamic therapy. Free Radic Biol Med. 2010;48:1296-1301.
- Mikolajewska P, Donnelly RF, Garland MJ, Morrow DI, Singh TR, Iani V, Moan J, Juzeniene A. Microneedle pre-treatment of human skin improves 5-aminolevulininc acid (ALA) and 5-aminolevulinic acid methylester (MAL)-induced PpIX production for topical photodynamic therapy without increase in pain or erythema. Pharm Res. 2010;27:2213-2220.
- Sakamoto FH, Torezan L, Anderson RR. Photodynamic therapy for acne vulgaris: a critical review from basics to clinical practice: part II. Understanding parameters for acne treatment with photodynamic therapy. J Am Acad Dermatol. 2010;63:195-211.
- Sasaki K, Watanabe M, Tanaka T, Tanaka T. Biosynthesis, biotechnological production and applications of 5-aminolevulinic acid. Appl Microbiol Biot. 2002;58:23-29.
- Edwards S, Jackson D, Reynoldson J, Shanley B. Neuropharmacology of δ-aminolaevulinic acid. II. Effect of chronic administration in mice. Neurosci Lett. 1984;50:169-173.
- Yang X, Li W, Palasuberniam P, Myers KA, Wang C, Chen B. Effects of silencing heme biosynthesis enzymes on 5‐aminolevulinic acid‐mediated protoporphyrin IX fluorescence and photodynamic therapy. Photochem Photobiol. 2015;91:923-930.
- Kannangara CG, Söll D, Gough S, Krupp G, Schön A, Berry-Lowe S. The RNA required in the first step of chlorophyll biosynthesis is a chloroplast glutamate tRNA. Nature. 1986;322:281-284.
- Lopez PJ, Guillerez J, Sousa R, Dreyfus M. On the mechanism of inhibition of phage T7 RNA polymerase by lac repressor. J Mol Biol. 1998;276:861-875.
- Maser A, Peebo K, Nahku R. Avoiding amino acid depletion in a complex medium results in improved Escherichia coli BW25113 growth. Microbiology. 2019;165:37-46.
- Maser A, Peebo K, Vilu R, Nahku R. Amino acids are key substrates to Escherichia coli BW25113 for achieving high specific growth rate. Res Microbiol. 2020;171:185-193.
- Wang H, La Russa M, Qi LS. CRISPR Cas9 in genome editing and beyond. Annu Rev Biochem. 2016;85:227-264.
- Khazen S, Molenaar C, Wiesmeijer K, Verwoerd NP, Eils R, Tanke HJ, Dirks RW. Visualizing telomere dynamics in living mammalian cells using PNA probes. EMBO J. 2003;22:6631-6641.
- Peng R, Lin G, Li J. Potential pitfalls of CRISPR/Cas9-mediated genome editing. FEBS J. 2016;283:1218-1231.
- Hsu PD, Scott DA, Weinstein JA, Ran FA, Konermann S, Agarwala V, Li Y, Fine EJ, Wu X, Shalem O, Cradick TJ, Marraffini LA, Bao G, Zhang F. DNA targeting specificity of RNA-guided Cas9 nucleases. Nat biotechnol. 2013;31:827-832.
- Phelan RM, Sachs D, Petkiewicz SJ, Barajas JF, Blake-Hedges JM, Thompson MG, Reider Apel A, Rasor BJ, Katz L, Keasling JD. Development of next generation synthetic biology tools for use in Streptomyces venezuelae. ACS Synth Biol. 2017;6:159-166.
- Burnham BF. δ-Aminolevulinic acid synthase (Rhodopseudomonas sphaeroides). Methods Enzymol. 1970;17A:195-204.
|
Table 1: Primer pairs for CRISPR/Cas9 induced Knock-In |
|||
|
Primer |
Sequences (5'to3') |
Base Number |
Note |
|
Primer F1 |
gcttgttggtacgcttctgccattcatccgcttattatcac |
41 |
Positions in boldface type are homologous
fragments designed for Takara In-Fusion |
|
Primer R1 |
accgcggcgtgggtacctggcgttacccaacttaat |
36 |
|
|
Primer ybhC-F |
tacccacgccgcggttattg |
20 |
|
|
Primer ybhC-R |
aagcgtaccaacaagcgcca |
20 |
|
|
Primer F2 |
cttttcgtgcgcgcataacgttgttaatctgtacctggtc |
40 |
Positions in boldface type are homologous
fragments designed for Takara In-Fusion |
|
Primer R2 |
tggtgtccgggatctgtatcgtttctggtcgcggcg |
36 |
|
|
Primer RNAP-F |
cagatcccggacaccatcga |
20 |
|
|
Primer RNAP-R |
atgcgcgcacgaaaagcatc |
20 |
|
|
Primer Donor-F |
tacccacgccgcggttattg |
20 |
|
|
Primer Donor-R |
aagcgtaccaacaagcgcca |
20 |
|
|
Primer N20-F |
gttttagagctagaaatagcaagttaaaat |
30 |
Embellished by 5’-phosphorylation |
|
Primer N20-R |
tctggaaacgaatcgtcagcactagtattatacctaggac |
40 |
|
|
|
Table 2. Strains and plasimds employed in this study |
|
|
Strain or plasmid |
Relevant characteristic(s) |
Source and/or reference |
|
Strain |
|
|
|
E. coli DH5α |
F-, φ80dlacZ ΔM15,Δ(lacZYA -argF )U169, deoR , recA1 , endA1 ,hsdR17
(rK-, mK+), phoA, supE44 , λ-, thi-1, gyrA96 , relA1 |
TransGen, Beijing |
|
E. coli BL21(DE3) |
fhuA2 [lon] ompT gal (λ DE3) [dcm] ΔhsdS λ DE3 = λ sBamHIo ΔEcoRI-
B int::(lacI::PlacUV5::T7 gene) i21 Δnin5 |
TransGen, Beijing |
|
E. coli BW25113 |
F-, DE(araD-araB)567, lacZ4787(del)::rrnB-3, LAM-, rph-1, DE(rhaD-
rhaB)568, hsdR514 |
Laboratory stock |
|
E. coli BW25113-T7 |
BW25113 int::(lacI::PlacUV5::T7 gene) ΔybhC |
This study |
|
Plasmids |
|
|
|
pACYCD-Blank |
E. coli cloning vector (p15a ori; CmR) |
Laboratory stock |
|
pACYCD-sYFP |
pACYCD-Blank with sYFP gene, LacI gene and T7-LacI promoter |
Laboratory stock |
|
pCas |
plasmid for CRISPR (temperature sensitive oriR101; KanR; the λ-Red
operon; S. pyogenes-derived cas9; sgRNA) |
Laboratory stock |
|
pTarget |
plasmid for CRISPR (p15a ori; CmR; sgRNA) |
Laboratory stock |
|
pTarget-ybhC |
plasmid for CRISPR (p15a ori; CmR; sgRNA guided to ybhC gene) |
This study |
|
pACYCD-ybhC |
pACYCD-Blank with ybhC gene (Fragment A) |
This study |
|
pACYCD-Donor DNA |
pACYCD-Blank with Donor DNA |
This study |
|
pACYCD-sYFP-17A (and so on) |
pACYCD-sYFP with T7 Promoter Variants Library |
This study |
|
pET28b-ALA-LA |
plasmid for biosyntheizing ALA (f1 ori; KanR; hemA; hemL; LacI gene
and T7-LacI promoter) |
Laboratory stock |
|
pET28b-ALA-LAR |
plasmid for biosyntheizing ALA (f1 ori; KanR; hemA; hemL; RhtA; LacI
gene and T7-LacI promoter) |
Laboratory stock |
- FigureS1.pdf
Figure S1. Confirmation of CRISPR/Cas9-mediated DSB in E. coli BW25113 The macroscopic images of colony formation after co-electroporation of Donor DNA and pTarget-ybhC.
- FigureS2.pdf
Figure S2. Detection of successful RNAP Knock in clones of BW25113-T7 A) The location of colony PCR product in BW25113-T7 genome. B) Lane M, 1kb Plus DNA ladder (TransGen, Beijing); lanes 1 was blank; lanes 2 to 8 were samples of colony PCR; lanes 8 and 9, positive control and negative control, respectively.
- TableS1PrimersforTestingandSequencing.xlsx
- TableS2PrimersforT7PromoterVariantsLibrary.xlsx
Journal Publication
published 12 Aug, 2021
Read the published version in Journal of Biological Engineering →
Editorial decision: Major revision
19 Apr, 2021
Review #3 received at journal
18 Apr, 2021
Review #1 received at journal
17 Apr, 2021
Reviewer #3 agreed at journal
03 Apr, 2021
Review #2 received at journal
02 Apr, 2021
Reviewer #2 agreed at journal
22 Mar, 2021
Reviews received at journal
22 Mar, 2021
Editor assigned by journal
21 Mar, 2021
Reviewers invited by journal
21 Mar, 2021
Reviewer #1 agreed at journal
21 Mar, 2021
Submission checks completed at journal
21 Mar, 2021
Editor invited by journal
21 Mar, 2021
First submitted to journal
16 Mar, 2021
You are reading this latest preprint version