Adding gentamicin to silk fibroin enhances both the antibacterial performance and degradation rate of silk-based materials. The increased material degradation rate can affect the strength of early internal fixation, resulting in internal fixation failure. This study sought to adjust the gentamicin concentration to control the material degradation rate, thereby better meeting clinical application requirements. The in vitro degradation, water absorption rate, and expansion rate of silk-based materials containing different gentamicin concentrations were studied. A gentamicin-loaded silk-based screw (GSS) was implanted into the femurs of New Zealand rabbits. Micro-computed tomography (Micro-CT) was used to measure the screw diameter, which was then used to calculate the degradation rate. The in vitro results revealed increasing material degradation rates with increasing gentamicin concentration but no significant differences in water absorption rates with different gentamicin concentrations. The degradation rates of gentamicin-loaded (4 mg/g) silk-based rod-like materials were approximately 11.08% at three months in vitro and 9.4% in the animal experiment. The time for complete degradation was predicted from the fitting curve to be approximately 16 months. The degradation of material containing 4 mg/g gentamicin meets clinical application requirements, and previous experimental results demonstrate good antibacterial performance is retained by materials containing this gentamicin concentration.
Research Article
Degradation of Internal Fixation Materials Based on Antibacterial and Absorbable Silk Containing Different Gentamicin Concentrations
https://doi.org/10.21203/rs.3.rs-672105/v1
This work is licensed under a CC BY 4.0 License
Journal Publication
published 22 Apr, 2022
Read the published version in Journal of Biomaterials Applications →
Version 1
posted
You are reading this latest preprint version
Absorbable materials are a future development trend for orthopedic internal fixation materials. Resorbable fixation devices of differing synthetic polymers, including polyglycolic acid (PGA) and poly-lactic-co-glycolic acid (PLGA), are often limited due to poor biocompatibility and potentially harmful degradation products1–3. Silk fibroin is a naturally renewable polymer and has confirmed biodegradability, biocompatibility, and impressive mechanical features4–8. Inprevious studies, gentamicin was added to silk-based materials to produce a novel degradable and antibacterial internal fixation material9. Its good biomechanical properties and cytocompatibility make this material an ideal candidate for an orthopedic internal fixation material. However, the degradation rate of gentamicin-loaded silk-based materials was significantly higher than that of the pure silk-based material. The in vitro degradation rate of gentamicin-loaded (16 mg/g) silk-based sheets was approximately 38% at three months, and the time for complete degradation time was predicted to be approximately five months9. The degradation time is critical for the clinical application of a novel internal fixation material. As the typical fracture healing time is approximately three months10, the rapid degradation of the internal fixation material within this period can affect the fixation strength and cause internal fixation to fail11–14. The degradation of silk protein is easily affected by environmental factors and synthesis process7,15,16. Therefore, the effect of different concentrations of added gentamicin on the material degradation rate was investigated. The gentamicin concentration was controlled to change the in vivo material degradation rate and therefore improve its clinical application prospects.
To predict the in vivo degradation of the silk-based internal fixation materials containing different gentamicin concentrations, the rod-like samples were slowly degraded in vitro by soaking in protease XIV, and the weight change was recorded. Protease XIV effectively decomposes silk fibroin fibers8. Figure 1 shows the in vitro degradation rates of the samples withdrawn at different time points. The silk-based internal fixation materials with four different gentamicin concentrations began to degrade after one week (p < 0.05). The degradation rates of GSS1, GSS2, GSS3, GSS4, and PSS were 2.91%, 2.13%, 1.45%, 1.04%, and 0.45%, respectively. The degradation continued with time. The degradation rates of GSS1, GSS2, GSS3, GSS4, and PSS were 19.71%, 14.12%, 13.03%, 11.08%, and 6.36% after 12 weeks, respectively. The degradation curves of the four antibacterial silk-based materials could all be fit with a third-order polynomial curve, whereas PSS could be fit with a second-order polynomial curve. The degradation rates at all time points decreased in the order of GSS1 > GSS2 > GSS3 > GSS4 > PSS (p < 0.05).
Figures 2a and b show the in vitro changes in the water absorption and expansion rates of the samples, respectively. The water absorption rates of GSS1, GSS2, GSS3, GSS4, and PSS were 1.91%, 1.91%, 1.89%, 1.88%, and 3.61%, respectively, and the expansion rates were 1.67%, 1.63%, 1.34%, 1.51%, and 1.96%, respectively. The results showed clear increases in the weight and diameter of the samples after 10 min (p < 0.05), indicating the onset of water absorption. However, the PSS diameter did not change significantly within 30 min and the GSS diameter did not increase significantly within 1 h (p > 0.05). After 48 h, the water absorption and expansion rates of all the samples tended to stabilize and did not increase further. At this time point, the water absorption rates of GSS1, GSS2, GSS3, GSS4, and PSS were 20.49%, 20.29%, 20.01%, 19.38%, and 30.69%, respectively, and the expansion rates were 9.50%, 9.32%, 8.78%, 9.12%, and 17.90%, respectively. There were no significant differences (p > 0.05) at each time point in the water absorption and expansion rates of the four antibacterial silk-based materials, though all were significantly smaller than those of PSS (p < 0.05).
All six rabbits were active after the operation, and no deaths occurred. Dissection was carried out after GSS4 had been implanted in the femur of the New Zealand rabbits for one month (Fig. 3a), two months (Fig. 3c), and three months (Fig. 3e). The screws were firmly fixed on the femurs and had rough surfaces. Although a partial defect on the nut on the surface of one femur was observed at three months, the main body of the screw in the femur remained intact and did not break. The screw was in close contact with the bone cortex, and the screw surface was covered with a large quantity of soft tissue. No adverse reactions (SEs), such as redness or inflammatory hyperplasia, were observed around the screw. The Micro-CT scans taken at one month (Fig. 3b), two months (Fig. 3d), and three months (Fig. 3f) showed that the screws were positioned well in the femurs without fracture or detachment, and no discernible adverse response was found in the surrounding tissues. The screw threads had clearly become shallow, the screw surface was rough, and high-density bone substances were observed around the screw. The rate of change in the GSS4 diameters in the New Zealand rabbits at one, two, and three months were 2.8%, 6.1%, and 9.4%, respectively (Fig. 4).
Degradation is very important for absorbable internal fixation materials. Excessively fast in vivo material degradation can affect the mechanical strength of the material and cause internal fixation to fail 8,17. Excessively slow degradation can result in the material remaining in the body for a long time. Previous experiments have shown that a sheet of the GSS1 material degraded significantly faster than PSS. The in vitro degradation rate of the GSS1 sheet was approximately 38% after three months, and the predicted time for complete degradation was approximately five months9. The healing time of most fractures in clinical applications is at least three months10,18. Therefore, GSS1 does not likely provide sufficient strength support during the first three months of fixation because of rapid degradation, causing the internal fixation to fail19. As gentamicin addition significantly increases the degradation rate of a silk-based material, the gentamicin concentration was varied in this study to control the material degradation rate.
In this study, four silk-based materials with different gentamicin concentrations and a pure silk-based rod-like material were soaked in a protease solution to simulate the degradation process. The degradation rate increased with the gentamicin concentration, where the degradation rates of all the gentamicin-loaded materials were higher than those of the pure silk-based material. The fitting curves (Fig. 1) were used to predict that the time for complete degradation of GSS1, GSS2, GSS3, GSS4, and PSS as approximately five, eight, 12, 16, and 29 months, respectively. This result confirmed previous experimental results that gentamicin addition accelerated the degradation rate of a silk-based material. We hypothesize that the dissolution of gentamicin particles increases the contact area between a material and an enzyme solution. Therefore, the higher the gentamicin concentration is, the larger the contact area between silk fibroin dissolved by gentamicin and the solution, and the higher the degradation rate19.
The healing time of a fracture in clinical applications is generally approximately three months, during which time the mechanical support provided by internal fixation is very important10,18. The in vitro degradation rate of GSS4 was approximately 11% after three months. A Micro-CT scan of the femur of a New Zealand rabbit implanted with GSS4 for three months was used to indirectly calculate a GSS4 degradation rate of 9.4%. Although this rate was close to the in vitro degradation rate, the in vivo degradation rate may be slower than that measured in the vitro simulation. Approximately 10% degradation after three months would result in a very low failure possibility of internal fixation. The dissection of the New Zealand rabbit after three months confirmed that the screw continued to provide solid support up to this time. The in vivo and in vitro experimental results were used to predict that the time for complete degradation as approximately 1.5 years, which meets the recommended removal time of internal fixation of 1.5-2 years18. Results from previous studies have shown that although the gentamicin concentration in GSS4 is lower than that in GSS1, the antibacterial rate of GSS4 against Staphylococcus aureus and Escherichia coli reached 100% after 6 h, indicating that 4 mg/g gentamicin retained effective antibacterial activity9. The degradation of GSS4 may satisfy clinical application requirements more effectively than GSS1, without a discernible deterioration in the antibacterial effect.
Water absorption is an important property of degradable internal fixation materials that also affects their placement and strength. The absorption and expansion rates of the four silk-based materials with different gentamicin concentrations were not significantly different and all smaller than those of the pure silk-based material. These findings were consistent with our previous results9. In vivo water absorption and expansion can increase the pullout resistance of internally fixed screws, thus increasing the internal fixation effect. However, an excessively rapid change in the screw diameter after exposure to water could increase the difficulty of internal fixation implantation. In this study, the change in the diameter of the antibacterial silk-based screws was significantly lower than that of the pure silk-based screw. The GSS diameter did not increase significantly within 1 h, which is sufficient time for a surgeon to implant GSSs.
The degradation rates of GSS1 and PSS were clearly lower in this study than the previous experimental study. The only difference between this study and the previous study is that rod-like materials were used in contrast to the sheet materials used in the previous study. The degradation rate of silk fibroin is affected by many factors, such as the shape of materials, the structure and diameter of silk fibroin fibers, and the implantation environment5,7. The following reasons may explain the aforementioned differences in the experimental results. As sheet material has a light weight and a small volume, the weight change from material degradation is relatively small. The small initial weight may significantly increase the experimental error in calculating a percentage value. The rod-like material is cylindrical and resembles a screw. Thus, the rod-like material reflects the in vivo degradation of a screw more accurately than a sheet.
Implanting GSS4 into the femurs of the New Zealand rabbits did not affect their normal activity, and the rabbits showed no morbidity. However, noticeable bone growth around the screw was observed by Micro-CT at one month, indicating good biocompatibility for GSS4. The screw surface was roughened by degradation, and stress was gradually transferred to the bone tissue as the fixation material slowly degraded. Thus, stress shielding was prevented, which may promote bone growth20–22. Animal experiments with a longer duration are required to explore the time for complete degradation, compatibility, and changes in the mechanical strength after degradation. In addition, the CT signal from the antibacterial silk-based material is near that of soft tissue, making it difficult to distinguish between these two materials. The preparation process of antibacterial silk-based materials needs to be further developed to obtain materials with clear CT signals and therefore promote clinical application.
The addition of gentamicin to a silk-based material significantly increased the degradation rate. The degradation rate increased with the gentamicin concentration. The in vitro degradation rate of a gentamicin-loaded (4 mg/g) silk-based rod-like material was approximately 11% at three months. The rate of change in the diameter of a GSS4 implanted into the femur of a New Zealand rabbit for three months was approximately 9.4%, which is relatively consistent with the results of the in vitro degradation experiment. The predicted time for complete GSS4 degradation was approximately 1.5 years. We conclude that in vivo degradation of GSS4 can meet actual clinical needs more effectively than that of GSS1, for which the predicted complete degradation time was approximately five months.
Materials: Cocoons were purchased from the Shandong Institute of Sericulture. Gentamicin was purchased from the National Institutes for Food and Drug Control. Protease Type XIV from Streptomyces griseus was purchased from Sigma-Aldrich (St. Louis, MO, USA), and other inorganic analytical reagents were purchased from Shanghai Aladdin.
Preparation of internal fixation materials: A gentamicin-loaded silk-based material was prepared by a previously reported method8,9. The cocoon was boiled in 0.02 mol/L sodium carbonate solution for 30 min, washed thoroughly with distilled water three times and placed in an electro-thermostatic blast oven at 60°C for at least 12 h to yield refined silk. A mass of 10 g of the refined silk was added to a ternary solution of CaCl2•CH3CH2OH•H2O (in a 1:2:8 molar ratio). The solution was heated to maintain the temperature at 85°C until the silk dissolved completely. The solution of the dissolved silk fibroin was placed in a dialysis bag (MWCO 3,500, Pierce) and dialyzed with deionized (DI) water at 4°C for 2 d to yield a pure silk fibroin solution. The DI water was changed hourly during the dialysis. The silk fibroin solution was centrifuged twice (18,000 rpm; 20 min/time) in a high-speed low-temperature centrifuge (4°C). The supernatant was collected and frozen at -20°C for 5 d and dried in a vacuum freeze dryer (-79°C). Gentamicin sulfate was dissolved in hexafluoroisopropanol (HFIP) at four concentrations (40, 20, 15, and 10 mg/ml), and pure HFIP was also prepared. The vacuum-dried silk (10 g) was cut into pieces and placed in a 50-ml syringe. A volume of 40 ml of the prepared HFIP solution was added to the syringe, and complete dissolution of the silk fibroin at room temperature yielded a 25 w/v% silk fibroin/HFIP solution. The solution was poured into a rod-shaped mold made of paraffin (a hollow column with a height of 33 mm and an inner diameter of 8 mm). The mold was completely immersed in a methanol solution for 3–4 d. A methanol-to-water gradient was carried out four times at 1-h intervals to gradually replace 100% methanol with 100% water. The rod-like material was left immersed in the water for an additional 2 d and then removed and dried in an oven at 60°C. Rods with a height of approximately 30 mm and a diameter of approximately 5 mm were thus obtained. Some of the rods were processed using a professional lathe to fabricate screws. Four gentamicin-loaded silk-based antibacterial materials (with gentamicin concentrations of 16 mg/g (GSS1), 8 mg/g (GSS2), 6 mg/g (GSS3), and 4 mg/g (GSS4)) and a pure silk-based material (PSS) were thus obtained.
In vitro degradation: Five rod-like samples (approximately 5 mm in diameter and 15 mm in height) were completely dried and weighed (the weight is denoted as m0). The samples were placed in a six-well plate (one sample per well), and 5 ml of a 1 mg/ml protease XIV solution were added to each well to completely immerse the samples (n = 3). At 37°C, the soaking solution was discarded every 48 h, the sample was washed with phosphate-buffered saline (PBS), and fresh protease XIV solution was added to the sample. At 7, 14, 28, 56, and 84 d, one sample was removed, washed thoroughly with PBS, dried thoroughly, and weighed (the weight was denoted as m1). The degradation rate (%) was calculated as 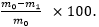
In vitro water absorption: The diameters and weights of five completely dried rod-like samples (approximately 5 mm in diameter and approximately 15 mm in height) were obtained (n = 3) and denoted by Wd and Dd, respectively. One rod-like sample was placed in an Eppendorf (EP) tube, 3 ml of PBS were added to immerse the material, and the tube was maintained at 37°C. Samples were withdrawn at predetermined time points (10 min, 20 min, 30 min, 1 h, 2 h, 6 h, 12 h, 24 h, 48 h, 72 h, 96 h, 120 h, and 144 h), filter paper was immediately used to absorb moisture on the sample surface, and the sample diameters and weights were measured and denoted by Ws and Ds, respectively. The water absorption and expansion rates were calculated using the following formulas: water absorption rate 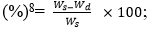 expansion rate (%) =
expansion rate (%) =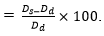
Animal experiments: Six New Zealand rabbits were numbered, weighed, and injected with 3% pentobarbital for anesthetization. The skin of each rabbit was prepared by removing the hair on both hind legs. The skin of one hind leg was cut open, and a retractor was used to pull the muscles away to expose the femoral condyle. An electric drill was used to drill and tap the femoral condyle from outside to inside, the GSS4 antibacterial silk-based screw (with a tail diameter of 5 mm, a body diameter of 4 mm and a length of 10 mm) was implanted, and the wound was sutured. The other hind leg of the rabbit was treated the same way. Two New Zealand rabbits were killed at 4, 8, and 12 weeks after the operation, and the four lower limbs were scanned by microcomputed tomography (Micro-CT) at each time point. Finally, the femur was dissected to observe the screw and surrounding tissue conditions. Micro-CT was used to measure the body diameter (d) (n = 4) of the screw in the femur at different time points, and the rate of change in the diameter (%) was calculated as 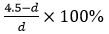 (the body diameter of the screw of 4.5 mm did not change after being soaked in a PBS solution for 48 h).
(the body diameter of the screw of 4.5 mm did not change after being soaked in a PBS solution for 48 h).
All experimental protocols were approved by Foshan First People's Hospital licensing committee and all methods were carried out in accordance with relevant guidelines and regulations. The study was carried out in compliance with the ARRIVE guidelines.
Statistical analysis: All the experimental data are expressed as the mean ± standard deviation, based on the results for at least three experimental samples. SPSS 20.0 software (SPSS Inc., Chicago, IL, USA) was used to perform statistical analysis. The one-way analysis of variance (ANOVA) and Student-Newman-Keuls (SNK) tests were performed for comparative analyses among multiple groups, and Dunnett’s T3 was used to compare the heterogeneity of variance. The significance level of the test was defined as α = 0.05, and differences were considered statistically significant at p < 0.05.
- Zheng, Z., Wu, J., Meng, L., Wang, H. & Li, C. 3D Bioprinting of Self-Standing Silk-Based Bioink.
- Zhang, J., Ebraheim, N., Lausé, G. E., Xiao, B. & Xu, R. A Comparison of Absorbable Screws and Metallic Plates in Treating Calcaneal Fractures: A Prospective Randomized Trial. The Journal of Trauma: Injury, Infection, and Critical Care, 1, doi:10.1097/TA.0b013e3182231811 (2011).
- Campbell, C. A. & Lin, K. Y. Complications of rigid internal fixation. Craniomaxillofacial trauma & reconstruction 2, 41-47, doi:10.1055/s-0029-1202596 (2009).
- Kostag, M., Jedvert, K. & El Seoud, O. Engineering of sustainable biomaterial composites from cellulose and silk fibroin: Fundamentals and applications. International journal of biological macromolecules 167, 687-718, doi:10.1016/j.ijbiomac.2020.11.151 (2021).
- Vepari, C. & Kaplan, D. L. Silk as a Biomaterial. Progress in polymer science 32, 991-1007, doi:10.1016/j.progpolymsci.2007.05.013 (2007).
- Kundu, B., Rajkhowa, R., Kundu, S. C. & Wang, X. Silk fibroin biomaterials for tissue regenerations. Advanced drug delivery reviews 65, 457-470 (2013).
- Altman, G. H. et al. Silk-based biomaterials. Biomaterials 24, 401-416, doi:10.1016/s0142-9612(02)00353-8 (2003).
- Perrone, G. S. et al. The use of silk-based devices for fracture fixation. Nature communications 5, 3385, doi:10.1038/ncomms4385 (2014).
- Shi, C. et al. An antibacterial and absorbable silk-based fixation material with impressive mechanical properties and biocompatibility. Scientific Reports 6, 37418, doi:10.1038/srep37418 (2016).
- Frost, H. M. The biology of fracture healing. An overview for clinicians. Part I. Clinical Orthopaedics & Related Research 248, 294 (1989).
- A, L. L. et al. In vitro and in vivo degradation of porous poly(DL-lactic-co-glycolic acid) foams. Biomaterials 21, 1837-1845 (2000).
- Greenwald, D., Shumway, S., Albear, P. & Gottlieb, L. Mechanical comparison of 10 suture materials before and after in vivo incubation. Journal of Surgical Research 56, 372-377 (1994).
- Dunn, M. G., Avasarala, P. N. & Zawadsky, J. P. Optimization of extruded collagen fibers for ACL reconstruction. Journal of Biomedical Materials Research 27, - (2010).
- Lutz et al. Resorbable polymer fibers for ligament augmentation. Journal of Biomedical Materials Research (2001).
- Zhou, J. et al. In vitro and in vivo degradation behavior of aqueous-derived electrospun silk fibroin scaffolds. Polymer Degradation & Stability 95, 1679-1685 (2010).
- Wang, Y. et al. In vivo degradation of three-dimensional silk fibroin scaffolds. Biomaterials 29, 3415-3428 (2008).
- Hovis, W. D., Watson, J. T. & Bucholz, R. W. Biochemical and Biomechanical Properties of Bioabsorbable Implants Used in Fracture Fixation. Techniques in Orthopaedics 13, 123-129 (1998).
- Rüedi, T. P., Buckley, R. E. & Moran, C. G. AO Principles of Fracture Management. 39 (Thieme, 2007).
- Horan, R. L. et al. In vitro degradation of silk fibroin. Biomaterials 26, 3385-3393, doi:10.1016/j.biomaterials.2004.09.020 (2005).
- Weitzel, P. P., Richmond, J. C., Altman, G. H., Calabro, T. & Kaplan, D. L. Future direction of the treatment of ACL ruptures. Orthopedic Clinics of North America 33, 653-661 (2002).
- Mandal, B. B., Grinberg, A., Gil, E. S., Panilaitis, B. & Kaplan, D. L. High-strength silk protein scaffolds for bone repair. Proceedings of the National Academy of Sciences 109, 7699-7704 (2012).
- Claes, L. E. Mechanical characterization of biodegradable implants. Clinical materials 10, 41-46 (1992).
No competing interests reported.
Journal Publication
published 22 Apr, 2022
Read the published version in Journal of Biomaterials Applications →
Version 1
posted
You are reading this latest preprint version