Primary hepatic NEC is a rare tumor, with an incidence of 0.3% among all neuroendocrine neoplasm (NEN) and 0.46% among primary hepatic malignancies [7]. As the liver is the most frequent site of NEN metastases from other organs, a systemic search for the primary lesion is necessary when hepatic NEN was suspected, considering the rarity of PHNEC. In contrast, HCC, the most common liver malignancy, often coexists with other malignancies. The most common of these combinations is HCC with intrahepatic cholangiocarcinoma, accounting for 2.0%–3.6% of all primary hepatic malignancies [8].
Although the exact origin of PHNEC is unclear, two hypotheses have been proposed [9, 10]: (a) neuroendocrine cells in the intrahepatic bile-duct epithelium undergo malignant conversion and become PHNEC; and (b) PHNEC originates from stem cells that have dedifferentiated from other malignant hepatic cells and convert into neuroendocrine cells. The latter concept can explain the lesions with different carcinomas at one site, whereas the former can only explain the pathogenesis of single PHNEC tumors.
In the literature, these composite liver tumors are classified as either collision type or combined type. In collision-type tumors, the HCC and NEC grow as separate microscopically-distinct compartments. In combined-type tumors, the HCC and NEC are closely intermingled and a transition zone can be found. These tumors used to be categorized as “mixed adeno-neuroendocrine carcinoma (MANEC)” when at least 30% of either component is identified [10]. However, with the revision of the World Health Organization (WHO) Classification in 2019, “collision type” tumors, in which two components appear to be independently derived and are simply adjoined with no observable mutual transition, were excluded from “mixed neuroendocrine neoplasm (MiNEN)” [11].
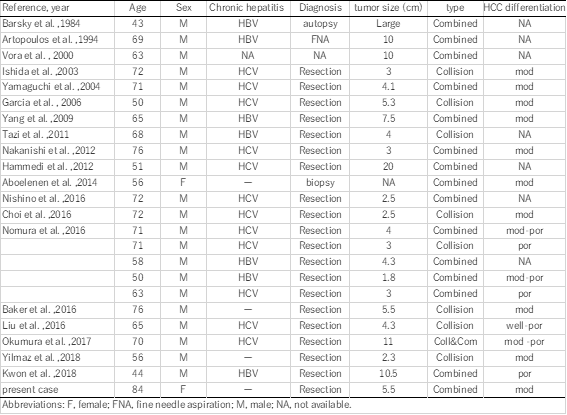
Concurrent occurrence of HCC and NEC is extremely rare. Including our patient, 21 English-language reports of 25 patients with HCC with NEC components have been published to date. The Clinicopathological profiles of these 25 patients are summarized in Table 1. These composite tumors were described as either collision type or combined type. In the present case, the two components were partly intermingled, and some portion of the HCC expressed neuroendocrine markers. Therefore, our case was classified as a combined-type MiNEN.
In the literature, combined type (n=17) tumors are more common than collision type (n= 9; one patient had both collision- and combined-type tumors). Almost all the reported cases were preoperatively diagnosed as HCC and re-diagnosed as combined or collision PHNEC–HCC tumors after resection, with only three cases diagnosed without surgery (by biopsy, fine needle aspiration, or autopsy); this demonstrates the difficulty of obtaining a correct preoperative diagnosis for these tumors.
In retrospect, the present case showed some interesting conformity between the preoperative images and the pathology. In the preoperative EOB-MRI, the lower and peripheral parts of the tumor showed a low signal in T1-weighted in- and out-of-phase images, indicating that this part contained fatty tissue and was thus highly suspicious of HCC. Furthermore, the upper and central parts showed extremely high signal intensity in the diffusion-weighted image (typical of NEC expression) just coinciding macroscopically with the distribution of HCC and NEC components in the tumor cut-surface, reflecting their mixture. Furthermore, the distributions of these components were morphologically presented as if the central part of the tumor had changed its character and had grown out to the ventral periphery. Previously, Yang et al. reported a combined-type tumor in which a poorly differentiated HCC focally expressed neuroendocrine marker [18], which resembled our case. In our case, these findings seem to support the supposition that a moderate or poorly differentiated HCC transdifferentiated into a neuroendocrine phenotype, resulting in a combined HCC–NEC tumor.
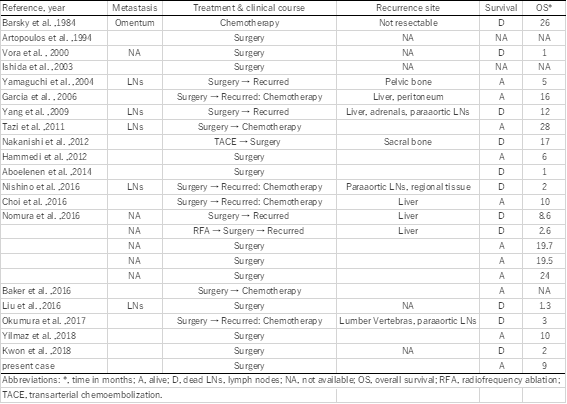
The clinical significance of HCCs with NEC components is unclear. Several reports have shown that HCCs with NEC components are associated with aggressive behavior and dismal outcomes [4,5]. Mixed PHNEC and HCC lesions tend to have a poor prognosis. Of the 25 cases summarized in this report, eight patients had recurrence, six patients died within a year after their surgeries, and only two patients were reportedly alive 2 years after surgery (Table 1). Although the number of reported cases is relatively small, the 1-year cumulative survival rate of the patients was 53% in our literature review (Figure 4; prognosis not available for three patients). Among resected cases with recurrence or biopsy-confirmed metastasis, an NEC component was found in each case, which indicates that the NEC component behaved more aggressively than primary HCC, leading to a much poorer prognosis. Therefore, identifying the neuroendocrine component is important for assuring that the patients can receive proper treatment. In our case, although patient was at a high age of 84, her liver was normal and without viral hepatitis or cirrhosis. We considered that surgery was a better treatment than tumor ablation or transcatheter arterial embolization (TAE) because the lesion was single and curative resection can be easily achieved. As a result, it led to the histological diagnosis of this complex tumor.
In conclusion, mixed PHNEC and HCC tumors are extremely rare. Further accumulation of case reports is required to clarify the features, diagnostic details, and optimal therapy for combined PHNEC–HCC lesions.