This article presents a comparative study of using three different kinds of biopolymers (xanthan gum, sodium alginate, and gelatin), environmentally friendly materials, in stabilizing collapsible soil. Modified Proctor, single oedometer, unconsolidated undrained triaxial, and California bearing ratio (CBR) tests were conducted to estimate the engineering characteristics of the soil after being mixed with various biopolymer contents. Biopolymer effect on the collapse index was investigated using different mixing conditions (dry mixing and wet mixing). Additionally, X-ray diffraction (XRD) and scanning electron microscopy (SEM) tests were utilized to demonstrate the changes in the microstructure of the treated samples. The results showed that after mixing the soil with 4% xanthan gum content in the dry case, the collapse index decreased significantly from 13.48–1.27% and the cohesion stress increased from 47 to 144 kPa, resulting in the shear strength improvement. The results proved that the superiority of xanthan gum over sodium alginate and gelatin in shear strength improvement is approximately 38% more than sodium alginate and 72% more than gelatin at the same conditions, as well as reducing the collapse index by approximately 5% more than sodium alginate and 12% more than gelatin at the same conditions. The findings also indicated that biopolymers increase the unsoaked CBR value by about 147%, 128%, and 117% when the soil was mixed with a 3% concentration of xanthan gum, sodium alginate, and gelatin, respectively. The results of SEM and XRD studies also demonstrated the interaction between the fine-grained particles and the strings of the biopolymers.
Research Article
A comparative study of stabilizing collapsible soil using different types of biopolymers
https://doi.org/10.21203/rs.3.rs-1972593/v1
This work is licensed under a CC BY 4.0 License
Version 1
posted
You are reading this latest preprint version
Biopolymer
collapsible soil
compaction
collapse potential
shear strength
CBR
SEM: XRD
Gradual population growth and quick expansion in the building sector have created a demand for enhancing existent soils to fit the applications of geotechnical engineering (Hojati et al., 2018).
Problematic soils are defined as any improper soil for construction purposes or whose behavior varies as environmental conditions change. Collapsible soil is an unstable soil that is widely spread worldwide and mostly exists in semi-arid and arid regions. It is defined by its sudden volumetric decrease in response to growing humidity. The collapse potential depends on various parameters including density, moisture content, applied stress, soil composition, and voids ratio. It is commonly determined as the proportion of the change in the volume of the soil which is subjected to specific vertical stresses after and before water immersion (Cerato et al., 2009; Houston et al., 2001). Collapsible soil has high apparent shear strength when it is unsaturated. This strength can be due to clay particles which cover and bind the soil particles together, causing collapsible soil cohesiveness. Some soluble materials, like gypsum and calcium chloride, can also be in charge of apparent cohesiveness between the soil particles. Therefore, collapsible soil is a stable soil when it is unsaturated, but when it is immersed, the water causes loss of cementation among the particles, causing significant volumetric changes (Clemence & Finbarr, 1981).
Soil stabilization means enhancing soil mechanical characteristics in order to achieve some predetermined aims (Latifi et al., 2018). Stabilization can be accomplished by several methods, including mechanical, physical, biological, electrical, and chemical processes (Phummiphan et al., 2018). The most common conventional addition used to stabilize the soil is Portland cement (Hojati & Radlińska, 2017). Unlikely, large volumes of soil demand a large amount of cement in order to be stabilized, which causes some defects, especially from the environmental point of view. In particular, cement production causes large amounts of nitrogen oxide (NOx) and carbon dioxide (C\({\text{O}}_{2}\)) gases, as well as cement dust emissions, which poses another possible environmental issue (Chang, Im, et al., 2015). Cement production exhausts about 12–15% of the global total energy in the sector of industry and produces about 5–7% of the world’s C\({\text{O}}_{2}\) emissions, which mainly contribute to the climate change (Gutiérrez et al., 2017). Alternatives to traditional soil treatment and stabilization methods (i.e., chemical treatment and mechanical improvement) include processes (like microbial-induced calcite precipitation (MICP)) and eco-friendly materials (like biopolymers) (Mendonça et al., 2021).
Biopolymer is a sustainable and carbon-neutral substance that is renewable since it is manufactured from non-food crops, which are available forever. Consequently, the application of biopolymers for purposes of geotechnical engineering will result in a more permanent industry (Stupp & Braun, 1997). Many recent studies have focused on enhancing soil shear strength by using biopolymers. Much research has been conducted on biopolymers (like guar gum, xanthan gum, agar, and modified starches) to enhance soil behavior (Chang, Im, et al., 2015; Chen et al., 2013). Biopolymers have succeeded in enhancing the shear strength behavior with different improvement degrees depending on soil types, biopolymer contents, biopolymer type, and curing conditions. Biopolymers not only enhanced the shear strength but also reduced soil permeability (M. K. Ayeldeen et al., 2016; Khachatoorian et al., 2003; Martin et al., 1996).
The cost of the material for 0.5% xanthan gum used for the stabilization process decreased from $250 to $28 in the previous thirty years. Most prospective biopolymers are now highly priced because of the high purity that is necessary for food quality. This culinary grade purity is not required for geotechnical applications, so the biopolymer cost may be predicted to be cheaper (Mendonça et al., 2021). Although biopolymers significantly succeeded in enhancing the behavior of ordinary soils, using biopolymers to stabilize problematic soils is nearly nonexistent.
This study aims to investigate and estimate the behavior of three different kinds of biopolymers that are used to stabilize collapsible soil using various contents and different mixing methods (dry and wet mix). The main parameters used in this study include compaction characteristics, collapse index, shear strength, and CBR value.
2.1. Soil
Collapsible soil samples, selected from a site located in New Borg-Alarab City in Egypt, were used in this study. Grain size analysis and hydrometer tests were conducted on soil sample following ASTM D 2487. The resulting grain size distribution curve is shown in Fig. 1. The major part of the soil was fine-grained. Table 1 shows the physical indices and chemical compositions of the tested soil.
2.2. Biopolymers
Three biopolymers were investigated: xanthan gum, sodium alginate, and gelatin. These biopolymers were chosen because they are inexpensive in comparison to other biopolymers (M. Ayeldeen et al., 2017, 2018).
Xanthan gum is an anionic exocellular polysaccharide generated by aerobic sugar fermentation by the bacterium Xanthomonas campestris. Its major chain consists of a linear 1, 4-linked β-D- glucose backbone substituted every two units with a charged tri-saccharide side chain (Hassler & Doherty, 1990). Xanthan gum is manufactured by Rama Gum Industries (India) Co., Ltd.
Sodium alginate is a natural polysaccharide consists of α-L-guluronic and β-D-mannuronic acid bonded through a (1→ 4) linkage (George & Abraham, 2006). It is derived as a byproduct of iodine and mannitol extraction from Sargasso of brown algae or kelp. It forms a crosslinked grid structure of hydrogel when it interacts with water. A small quantity of sodium alginate considerably raises the liquid’s viscosity, under static circumstances. The hydrogel, generated by the interaction between sodium alginate and water, can preserve viscosity stability throughout a large range of temperatures (below 80°C) and pH (Pignolet et al., 1998; Shalumon et al., 2011). The investigated powder of sodium alginate came from a manufacturing company (Central Drug House (P) Ltd., India).
Gelatin is derived from collagen by thermal denaturalization, and in the existence of dilute acids, collagen is present in animal bones and skin. It has a significant amount of proline, glycine, and 4-hydroxyproline residues. It is transparent and approximately savorless powder. It is mainly utilized in pharmaceutical, food, and cosmetic products as a gelling agent, and it generates gel when cooled below 35°C. It exists in a collagen fold order that is able to generate hydrogen bonds at lower temperatures (Robinson, 1953). But it exists as a single molecule above 35°C, and it isn't able to form interchain hydrogen bonds. Chemical crosslinking can change gelatin gelling characteristics (Hayashi et al., 2007; Sutter et al., 2007).
2.3. Specimen preparation
Under laboratory conditions, the natural soil was air-dried. While drying, plant roots and pebbles were eliminated. In order to guarantee homogeneity, only soil that passed through a #40 sieve was used. The soil was then oven dried to eliminate all its moisture content before being mixed with the biopolymer. The soil was mixed with biopolymer using two methods: dry mix and wet mix. This study mainly depends on the dry mix method, so the specimens for all tests were prepared using the dry mix technique. In this technique, a certain mass percentage of biopolymer powder was thoroughly blended with the soil. Secondly, distilled water was weighted and mixed with the soil-biopolymer mixture in order to obtain a water content of 6.7% for specimens prepared at a dry density of 13.6 kN/\({\text{m}}^{3}\)and a water content of 12% for specimens prepared at a dry density of 16.3 kN/\({\text{m}}^{3}\), respectively, as shown in Table 2. It is necessary to use hand mixing with a palette knife to ensure the homogeneity of the mixtures. Here, the biopolymer content is determined as the mass of biopolymer powder (\({\text{m}}_{\text{b}}\)) to dry soil mass (\({\text{m}}_{\text{s}}\)), presented as a percentage (\({\text{m}}_{\text{b}}\) x100/\({\text{m}}_{\text{s}}\)). On the other hand, the wet mix procedure was used to prepare specimens for collapse index tests to investigate the effect of mixing methods on the collapse index, as it is the main issue of the collapsible soil. In this method, the solution of the biopolymer was first prepared by adding the powder to the water slowly, and the solution was stirred until homogeneity and then mixed with the dry soil. A homogeneous solution with specified biopolymer content was obtained by dissolving the powder in hot distilled water (85ºC) and mixing with the magnetic stirrer. After preparing mixtures, it was necessary to compact all specimens to obtain the same density before being tested. Therefore, the compaction characteristics was first investigated. According to compaction results, which will be explained later, the maximum dry density (\({{\gamma }}_{\text{d} \text{m}\text{a}\text{x}})\) of the mixtures varied as it decreased with increasing biopolymer content. Compaction results also showed that the highest \({{\gamma }}_{\text{d} \text{m}\text{a}\text{x}}\) was in the case of the untreated soil sample where it was 18.1 kN/\({\text{m}}^{3}\)and the least \({{\gamma }}_{\text{d} \text{m}\text{a}\text{x}}\) was in the case of 2% gelatin-soil sample where it was 16.2 kN/\({\text{m}}^{3}\)which represents about 90% of \({{\gamma }}_{\text{d} \text{m}\text{a}\text{x}.}\)of untreated soil. As a result, all specimens were prepared at 90% of \({{\gamma }}_{\text{d} \text{m}\text{a}\text{x}}\)of the untreated soil. As shown in Table 2, other specimens which were used in collapse index tests were prepared at 75% of \({{\gamma }}_{\text{d} \text{m}\text{a}\text{x}}\) of the untreated soil since the natural density was nearly 75% of \({{\gamma }}_{\text{d} \text{m}\text{a}\text{x}}\)of the untreated soil.
2.4. Test procedures and experimental program
The experimental program included compaction, single oedometer, unconsolidated-undrained (UU) triaxial, California bearing ratio (CBR), X-ray diffraction (XRD), and scanning electron microscopy (SEM) tests, which have been performed to determine the engineering characteristics and of the soil before and after treatment. Table 2 summarizes the laboratory schedule for these experiments.
In accordance with ASTM D1557-07, compaction tests were performed on soil–biopolymer mixtures to study the effect of adding various contents of biopolymers on the maximum dry density and optimum water content.
Single oedometer tests were carried out following ASTM D 5333 to study the collapse behavior. The vertical stress was steadily raised until it reached 200 kPa, at which point the specimen was immersed in water for 24 hours. According to the specification, the variance in axial strain (%) after and before immersing at a vertical stress of 200 kPa is determined to be the collapse index of the specimen. The failed specimens were collected after the test for compositional analysis and microscopic testing.
Shear strength characteristics of the specimens were determined through UU tests following ASTM D2850-87 where the specimens were subjected to three confining pressures of 50, 100, and 200 kPa. Loading was performed at a rate of 1% strain per minute with an automated device and continued until the strain reached 15%, unless the specimen's failure was earlier.
CBR tests were performed following ASTM D1883-99. CBR is determined as the ratio of the specimen's load at a penetration of 2.5- or 5.0-mm to the load sustained by standard load aggregates at the same penetration level. The unsoaked condition was used to test all of the specimens with different biopolymer contents.
XRD analysis was conducted to examine the microstructure and mineral composition of the soil before and after treatment. This technique is widely used to investigate new mineral presence in treated soil (Kong et al., 2018; Latifi et al., 2016; Rashid et al., 2017). A wide Bragg angle (2θ) range (4°–80°) and a rate of 0.02°/s were selected to investigate the mineral composition of the samples thoroughly.
SEM is a common technique for evaluating the microstructural characteristics of the soil. It scans the surface of the sample with a concentrated beam of electrons in order to obtain pictures. Electrons interact with sample’s atoms, producing a variety of signals. These tested samples were employed to notice the presence of new cementitious materials.
3.1. Compaction characteristics
It is necessary to compact the soil after combining with biopolymers to a certain density level as part of the primary process utilized to improve soil layers. Other mechanical characteristics, including bearing capacity, settling, and shear strength, will be influenced by the achieved density after compaction. Therefore, the compaction characteristics of collapsible soil blended with various biopolymer contents had to be studied. When the biopolymer content of gelatin, xanthan gum, and sodium alginate increased, the maximum dry density\(\left({{\gamma }}_{\text{d} \text{m}\text{a}\text{x}}\right)\) decreased and optimum water content (OWC) increased, as shown in Fig. 2 and Fig. 3. For gelatin-treated specimens, when the concentration was increased from zero to 2%, the density dropped from 18.1 to 16.2 kN/\({\text{m}}^{3}\). The reduction in dry density was less for sodium alginate and xanthan gum than that for gelatin, where \({{\gamma }}_{\text{d} \text{m}\text{a}\text{x}}\) reached 17.94 kN/\({\text{m}}^{3}\) and 16.7 kN/\({\text{m}}^{3}\) at 2% sodium alginate and 2% xanthan gum, respectively.
This behavior can be explained by the soil partial weight and the biopolymer solution viscosity as the light weight of soil particles allows them to move away from one another in response to the influence of the solution viscosity, resulting in a total decrease in dry density. Furthermore, the solution viscosity increases when the biopolymer content increases, which results in an even further reduction in the dry density of the soil.
On the other hand, optimum water content rose from 16.30% at zero content to 21.40%, 18.12%, and 21.80% at 2% content of xanthan gum, sodium alginate, and gelatin, respectively. This behavior is because increasing the biopolymer content results in an increase in the absorbed water used to dissolve biopolymer, which matches the trend observed by (M. Ayeldeen et al., 2017; M. K. Ayeldeen et al., 2016; Chang, Prasidhi, et al., 2015; Dehghan et al., 2019).
3.2. Collapse index
The collapse index has been studied for two cases of dry densities according to the experimental program as shown in Table 2. For specimens prepared with a dry density of 1.36 kN/\({\text{m}}^{3}\), the collapse index was assessed for two cases of mixing techniques: dry mix and wet mix. The collapse behavior was considerably influenced by biopolymer type, biopolymer content, and mixing technique. As shown in Fig. 4.a, the collapse index was reduced by increasing xanthan gum content in dry and wet mix cases, but it was observed that the wet mix case was superior to the dry mix case. Figure 5 presents the impact of xanthan gum content on the collapse test results in the case of wet mixing. Even at low concentrations, the xanthan gum content has a considerable effect on the collapse index, as the collapse index was reduced from 13.48–7.14% by adding 0.50% xanthan gum content. When xanthan gum content increased to 1%, the collapse index decreased to 4.19%. The collapse index was reduced to 0.3%, which is approximately vanished, at a content of 4%.
Figure 6 presents the collapse index test results for untreated specimens and treated specimens with 4% xanthan gum for the two mixing methods. The collapse index was 13.48% for untreated soil, while it was 1.27% and 0.30% for dry mix and wet mix cases, respectively. The efficiency of xanthan gum was around 90% in decreasing the collapse index of the soil for the dry mix case, while it was 98% for the wet mix case.
The effect of sodium alginate and gelatin on the collapse index was similar to xanthan gum effect and it was obvious that the wet mix was more effective than the dry mix, as shown in Fig. 4.b and Fig. 4.c. Gelatin is less efficient than sodium alginate, and all of them are less efficient than xanthan gum, as presented in Table 3 and Fig. 7.
In the wet mix method, about 2.15% xanthan gum, 3.12% sodium alginate, and 4% gelatin are necessary to reach the "Slight" stage of the collapse index. For the dry mix case, about 2.9% xanthan gum content is necessary to reach the same stage, about 4% sodium alginate, and more than 4% of gelatin are needed.
According to (Chang, Im, et al., 2015), biopolymers’ specific surfaces are quite high and electrically charged, allowing for direct interaction with clayey soil particles. According to the microscopic behavior of the sample, which was stabilized by 2% xanthan gum, SEM pictures show a direct interaction between xanthan gum and soil particles, as shown in Fig. 15 which is presented in SEM results. The electrical charge of fine-grained soil particles may motivate this interaction, in which xanthan monomers are linked to the electrically charged soil particles via hydrogen bonding and cationic bonding between hydroxyl groups (-OH) and carboxylic acids (-COOH), which is accompanied by the aggregation of particles and filling the voids with air or biopolymer gel. The superiority of xanthan gum solution as compared with sodium alginate solution and gelatin solution is due to its stronger hydrogen bonding and smaller voids (M. Ayeldeen et al., 2017; M. K. Ayeldeen et al., 2016; Chen et al., 2013).
The similar effect was obtained for specimens which were prepared at a dry density of \(1.63 \text{k}\text{N}/{m}^{3}\)as shown in Table 4. With increasing biopolymer content, the collapse index was reduced. With a content of 4% of xanthan gum, sodium alginate, and gelatin, the collapse index decreased to 0.40%, 0.50% and 1.71%, respectively.
3.3. Shear strength
Shear strength is a key parameter of soil improvement. The shear strength parameters of the tested samples were obtained from the unconsolidated undrained triaxial test by plotting the stress Mohr’s circle and shear strength envelope diagrams as presented in Fig. 8. Figure 9, Fig. 10, and Fig. 11 present failure envelopes for untreated specimens and treated specimens with different contents of biopolymers. In comparison with untreated specimens, all treated specimens exhibited a considerable increase in cohesion intercept and total shear strength, although the friction angle was reduced. Figure 12.a shows that after mixing the soil with 0.5% of xanthan gum, sodium alginate, and gelatin, the friction angle has been reduced from 34.34° (for untreated sample) to 34.08°, 33.9°, and 34.2°, respectively. Increasing the content from 0.5 to 4% of xanthan gum, sodium alginate, and gelatin slightly reduced the friction angle to 31.66°, 31.312°, and 32°, respectively. This reduction is because the biopolymer coating effect on the surfaces of the grains softened the microscale roughness, resulting in less asperity interlocking of sand grains. This resulted in an inconsiderable decrease in the friction angle (M. Ayeldeen et al., 2017).
As shown in Fig. 12.b, cohesion intercept has been increased from 47 for zero content to 144, 117, and 95 kPa for 4% content of xanthan gum, sodium alginate, and gelatin, respectively. This improvement is because biopolymers have numerous chemical functional groups, like amines, ester, or hydroxyl groups, whose extended chains offer additional sites for the distinguished chemical interactions of a mentioned functional group to take place. Chemical bonding is responsible for the adhesive forces that hold the soil particles and the surface of the gel together (Khatami & O’Kelly, 2013). On a microscopic scale, the kinds of forces among the gel and the particles influence the efficiency of the bonding. Ionic/electrostatic or covalent connections (chemisorption), hydrogen bonding (strong polar attraction), and van der Waals forces (physical absorption) all operate at the interface between the gel and the particles. Short-range covalent and ionic/electrostatic bonds give the strongest bonds because of their high bond energy. Van der Waals forces give the weakest bonds over a long range. On the other hand, the increase in molecular weight of the biopolymer results in an increase in the solution viscosity because the higher molecular weight of the biopolymer enhances the opportunity of sustaining its macromolecule chain crystallization, which results in an increase in the degree of crosslinking within the soil matrix. Thus, the xanthan gum mixture had a higher shear strength than sodium alginate mixture and gelatin mixture because xanthan gum solution had a higher viscosity than sodium alginate and gelatin, as stated before in (M. Ayeldeen et al., 2017; Dehghan et al., 2019).
Cohesion stress isn't the only parameter influencing the soil shear strength. The increase in the biopolymer content led to a slight reduction in the friction angle and an increase in the cohesion stress. So, to study the shear strength behavior after soil treatment, the overall shear strength of the soil was determined in accordance with the next equation:
\({\tau }_{f}\) = c + (σ + γ h) tan ϕ
where \({\tau }_{f}\) is the soil shear strength which is presented in Fig. 12.c, c is the cohesion stress, σ is the external stress, ϕ is the friction angle, and γh is the overburden pressure at depth h of 1.50 m below the ground.
3.4. California bearing ratio
CBR is an important parameter for pavement design (Abo El-Naga et al., 2020). This depends on the fact that higher CBR offers a lower risk of strain under loads. Additionally, CBR is an expression of the shear strength, bearing capacity, and resilience modulus of the soil (Abo El-Naga et al., 2020; Choudhary & Joshi, 2014). Since this study is concerned in soil stabilization for construction purposes or to be used as subgrade for pavements, the engineering characteristic of interest is the CBR. Unsoaked CBR tests were performed on untreated specimens and treated specimens with biopolymer contents of 1%, 2%, and 3%. It is noticed that CBR was significantly increased by increasing the biopolymer content, as shown in Fig. 13 and Fig. 14. The findings also revealed that the xanthan gum effect on the CBR value was more remarkable than sodium alginate and gelatin. CBR increased from 6.52% (for natural soil) to 8.22%, 11.14%, and 16.14% when the soil was mixed with contents of 1%, 2%, and 3% of xanthan gum, respectively. The increase in CBR value was less in the case of sodium alginate and gelatin specimens, as shown in Fig. 13 and Fig. 14. The unsoaked CBR value increased by about 147%, 128%, and 117% when the soil was treated with concentrations of 3% xanthan gum, sodium alginate, and gelatin, respectively.
3.5. Discussion
The previous findings revealed that xanthan gum, sodium alginate, and gelatin can considerably alter both the chemical composition and physical structure of the collapsible soil. Biopolymers contain a large number of -COO- groups which become polyanionic when dissolved in water, causing adhesion. Biopolymer can form a hydrogel when it is combined with water under mild conditions (Ouwerx et al., 1998; Zhao et al., 2020). The soil's microstructure is considerably changed by this hydrogel. SEM study was performed on a sample of soil with 2% xanthan gum content. From the findings of SEM study shown in Fig. 15, it has been observed that xanthan gum has three main impacts on the soil's microstructure: First, the holes among the soil particles were filled by the colloids generated by xanthan gum, which resulted in reducing the soil's porosity. As a result, the contact among the particles altered from point contact to surface contact, which caused an increase in the contact area while the structure of the soil was strengthened. Second, addition of xanthan gum formed a coating effect on the particles, which were encapsulated by a film-like gel substance that increased the particle size. As the particles became larger, they could touch each other easier. Furthermore, because of flocculation, the clay particles remained bonded together and aggregated to form larger particles.
Also, the findings of XRD analysis have proved the coating presence for the used biopolymers. Figure 16, 17, and 18 indicate that there is no presence of new crystalline substance in the stabilized soil as the main minerals were still calcite and quartz. Although no additional peaks appeared, the intensities of the initial peaks in the XRD pictures gradually reduced as the biopolymer content increased, and the curve tended to be gentle, as shown in Fig. 16, Fig. 17, and Fig. 18. The SEM pictures indicated that this is because the biopolymer hydrogel enclosed the crystals. The hydrogel coating on the crystal surface blocked X-rays from being directed into the soil crystals, which is the same trend presented by (Zhao et al., 2020).
Three kinds of biopolymers: xanthan gum, sodium alginate, and gelatin, were investigated to stabilize a natural collapsible soil, and these are the results which have been concluded:
- With increasing the biopolymer content from zero to 2% for xanthan gum, sodium alginate, and gelatin, the maximum dry density decreased from 18.1 kN/ to 16.7 kN/ 17.94 kN/ and 16.2 kN/ , and the optimum water content increased from 16.3% to 21.4%, 18.12%, and 21.8%, respectively.
- Xanthan gum, sodium alginate, and gelatin considerably reduced the collapse index for both wet and dry methods and for different densities because of filling the pores and particle bonding, which play an important role in stabilizing the collapsible soil.
- When the soil was mixed using the dry mix method with 4% of xanthan gum, sodium alginate, and gelatin, the collapse index had decreased from 13.48% to 1.27%, 2.05%, and 2.9%, respectively.
- When the soil was mixed using the wet mix method with 4% of xanthan gum, sodium alginate, and gelatin, the collapse index reached 0.3%, 0.95%, and 2%, respectively.
- Although increasing biopolymer content reduced the maximum dry density which led to a slight decrease in the friction angle, the shear strength was enhanced due to the significant increase in cohesion intercept.
- Cohesion intercept increased by 206%, 149%, and 102% when the soil was mixed with 4% of xanthan gum, sodium alginate, and gelatin, respectively.
- At a concentration of 4% xanthan gum, sodium alginate, and gelatin, the unsoaked CBR value of the soil was increased by 148%, 128%, and 117%, respectively.
- SEM micrographs showed the development of crosslinked components and hydrogels within the soil matrix due to the electrostatic bonding or hydrogen among the soil particles and biopolymer. These gel components are in charge of the improvement in collapse index, strength, and CBR value of untreated soil.
- Though no new crystalline substance was observed in XRD analysis for treated samples, the biopolymer hydrogel had a coating and filling impact that changed the microstructure of the collapsible soil.
- Xanthan gum is superior to sodium alginate and gelatin in improving soil behavior.
Acknowledgments
Mahmoud Habib is greatly thankful to his family especially his father, Eng. Saeed Habib, for his continuous help, encouragement, and endless support.
Author Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Mahmoud S. Habib. The first draft of the manuscript was written by Mahmoud S. Habib. Prof. Dr. Mostafa A. El Sawwaf, Prof. Dr. Marawan M. Shahien, and Prof. Dr. Ahmed M. Nasr commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding
Open access funding is provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB). The authors have no relevant financial or non-financial interests to disclose.
Data availability
All data, models, and code generated or used during the study appear in the submitted article.
Ethical approval Not applicable.
Consent to participate Not applicable.
Consent for publication Not applicable.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
- Abo El-Naga, I., Elsiragy, M. N., & Ragab, M. (2020). Utilization of synthetic reinforcement for enhancement of oil-contaminated subgrade soil in highway pavement. Innovative Infrastructure Solutions, 5(2), 1–10.
- Ayeldeen, M. K., Negm, A. M., & El Sawwaf, M. A. (2016). Evaluating the physical characteristics of biopolymer/soil mixtures. Arabian Journal of Geosciences, 9(5), 1–13.
- Ayeldeen, M., Negm, A., El-Sawwaf, M., & Kitazume, M. (2017). Enhancing mechanical behaviors of collapsible soil using two biopolymers. Journal of Rock Mechanics and Geotechnical Engineering, 9(2), 329–339. https://doi.org/10.1016/j.jrmge.2016.11.007
- Ayeldeen, M., Negm, A., El Sawwaf, M., & Gädda, T. (2018). Laboratory study of using biopolymer to reduce wind erosion. International Journal of Geotechnical Engineering, 12(3), 228–240. https://doi.org/10.1080/19386362.2016.1264692
- Cerato, A. B., Miller, G. A., & Hajjat, J. A. (2009). Influence of clod-size and structure on wetting-induced volume change of compacted soil. Journal of Geotechnical and Geoenvironmental Engineering, 135(11), 1620.
- Chang, I., Im, J., Prasidhi, A. K., & Cho, G.-C. (2015). Effects of Xanthan gum biopolymer on soil strengthening. Construction and Building Materials, 74, 65–72.
- Chang, I., Prasidhi, A. K., Im, J., & Cho, G.-C. (2015). Soil strengthening using thermo-gelation biopolymers. Construction and Building Materials, 77, 430–438.
- Chen, R., Zhang, L., & Budhu, M. (2013). Biopolymer stabilization of mine tailings. Journal of Geotechnical and Geoenvironmental Engineering, 139(10), 1802–1807.
- Choudhary, D. K., & Joshi, Y. P. (2014). A detailed study of Cbr method for flexible pavement design. International Journal of Engineering Research and Applications, 4(6), 239–253.
- Clemence, S. P., & Finbarr, A. O. (1981). Design considerations for collapsible soils. Journal of the Geotechnical Engineering Division, 107(3), 305–317.
- Dehghan, H., Tabarsa, A., Latifi, N., & Bagheri, Y. (2019). Use of xanthan and guar gums in soil strengthening. Clean Technologies and Environmental Policy, 21(1), 155–165. https://doi.org/10.1007/s10098-018-1625-0
- George, M., & Abraham, T. E. (2006). Polyionic hydrocolloids for the intestinal delivery of protein drugs: alginate and chitosan—a review. Journal of Controlled Release, 114(1), 1–14.
- Gutiérrez, A. S., Eras, J. J. C., Gaviria, C. A., Van Caneghem, J., & Vandecasteele, C. (2017). Improved selection of the functional unit in environmental impact assessment of cement. Journal of Cleaner Production, 168, 463–473.
- Hassler, R. A., & Doherty, D. H. (1990). Genetic engineering of polysaccharide structure: production of variants of xanthan gum in Xanthomonas campestris. Biotechnology Progress, 6(3), 182–187.
- Hayashi, K., Kubo, T., Doi, K., Tabata, Y., & Akagawa, Y. (2007). Development of new drug delivery system for implant bone augmentation using a basic fibroblast growth factor-gelatin hydrogel complex. Dental Materials Journal, 26(2), 170–177.
- Hojati, M., Nazarian, S., & Duarte, J. (2018). Seamless architecture: design and development of functionally-graded green materials for building construction. 4th Residential Building Design and Construction Conference (RBDCC), State College, PA.
- Hojati, M., & Radlińska, A. (2017). Shrinkage and strength development of alkali-activated fly ash-slag binary cements. Construction and Building Materials, 150, 808–816.
- Houston, S. L., Houston, W. N., Zapata, C. E., & Lawrence, C. (2001). Geotechnical engineering practice for collapsible soils. In Unsaturated soil concepts and their application in geotechnical practice (pp. 333–355). Springer.
- Khachatoorian, R., Petrisor, I. G., Kwan, C.-C., & Yen, T. F. (2003). Biopolymer plugging effect: laboratory-pressurized pumping flow studies. Journal of Petroleum Science and Engineering, 38(1–2), 13–21.
- Khatami, H. R., & O’Kelly, B. C. (2013). Improving mechanical properties of sand using biopolymers. ASCE J. Geotech. Geoenviron. Eng, 139.
- Kong, R., Zhang, F., Wang, G., & Peng, J. (2018). Stabilization of loess using nano-SiO2. Materials, 11(6), 1–14. https://doi.org/10.3390/ma11061014
- Latifi, N., Horpibulsuk, S., Meehan, C. L., Majid, M. Z. A., & Rashid, A. S. A. (2016). Xanthan gum biopolymer: an eco-friendly additive for stabilization of tropical organic peat. Environmental Earth Sciences, 75(9), 2–11. https://doi.org/10.1007/s12665-016-5643-0
- Latifi, N., Vahedifard, F., Ghazanfari, E., & Rashid, A. S. A. (2018). Sustainable usage of calcium carbide residue for stabilization of clays. J Mater Civ Eng, 30(6), 4018099.
- Martin, G. R., Yen, T. F., & Karimi, S. (1996). Application of biopolymer technology in silty soil matrices to form impervious barriers. Proceedings of 7 Th Australia New Zeeland Conference on Geomechanics, 814–819.
- Mendonça, A., Morais, P. V., Pires, A. C., Chung, A. P., & Oliveira, P. V. (2021). A review on the importance of microbial biopolymers such as xanthan gum to improve soil properties. In Applied Sciences (Switzerland) (Vol. 11, Issue 1, pp. 1–14). MDPI AG. https://doi.org/10.3390/app11010170
- Ouwerx, C., Velings, N., Mestdagh, M. M., & Axelos, M. A. V. (1998). Physico-chemical properties and rheology of alginate gel beads formed with various divalent cations. Polymer Gels and Networks, 6(5), 393–408.
- Phummiphan, I., Horpibulsuk, S., Rachan, R., Arulrajah, A., Shen, S.-L., & Chindaprasirt, P. (2018). High calcium fly ash geopolymer stabilized lateritic soil and granulated blast furnace slag blends as a pavement base material. Journal of Hazardous Materials, 341, 257–267.
- Pignolet, L. H., Waldman, A. S., Schechinger, L., Govindarajoo, G., & Nowick, J. S. (1998). The alginate demonstration: Polymers, food science, and ion exchange. Journal of Chemical Education, 75(11), 1430.
- Rashid, A. S. A., Latifi, N., Meehan, C. L., & Manahiloh, K. N. (2017). Sustainable Improvement of Tropical Residual Soil Using an Environmentally Friendly Additive. Geotechnical and Geological Engineering, 35(6), 2613–2623. https://doi.org/10.1007/s10706-017-0265-1
- Robinson, C. (1953). The hot and cold forms of gelatin. Nature and Structure of Collagen. Academic Press, New York, 96–105.
- Shalumon, K. T., Anulekha, K. H., Nair, S. V, Nair, S. V, Chennazhi, K. P., & Jayakumar, R. (2011). Sodium alginate/poly (vinyl alcohol)/nano ZnO composite nanofibers for antibacterial wound dressings. International Journal of Biological Macromolecules, 49(3), 247–254.
- Stupp, S. I., & Braun, P. V. (1997). Molecular manipulation of microstructures: biomaterials, ceramics, and semiconductors. Science, 277(5330), 1242–1248.
- Sutter, M., Siepmann, J., Hennink, W. E., & Jiskoot, W. (2007). Recombinant gelatin hydrogels for the sustained release of proteins. Journal of Controlled Release, 119(3), 301–312.
- Zhao, Y., Zhuang, J., Wang, Y., Jia, Y., Niu, P., & Jia, K. (2020). Improvement of loess characteristics using sodium alginate. Bulletin of Engineering Geology and the Environment, 79(4), 1879–1891. https://doi.org/10.1007/s10064-019-01675-z
Table 1 Physical properties and chemical compositions of the collapsible soil.
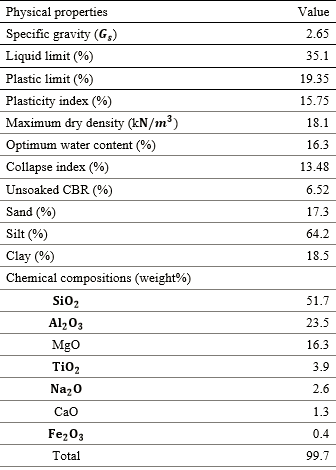
Table 2 Experimental program summary
|
Variable |
Modified proctor test |
Single oedometer test |
Unconsolidated undrained triaxial test |
California bearing ratio test |
|
|
Wet mix |
Dry mix |
||||
|
Xanthan gum content (%) |
0.5, 1, 1.5, 2 |
0.5, 1, 1.5, 2, 3, 4 |
0.5, 1, 2, 3, 4 |
1, 2, 3 |
|
|
Sodium alginate content (%) |
0.5, 1, 1.5, 2 |
0.5, 1, 1.5, 2, 3, 4 |
0.5, 1, 2, 3, 4 |
1, 2, 3 |
|
|
Gelatin content (%) |
0.5, 1, 1.5, 2 |
0.5, 1, 1.5, 2, 3, 4 |
0.5, 1, 2, 3, 4 |
1, 2, 3 |
|
|
Dry density (kN/m2) |
---------- |
13.6 |
13.6, 16.3 |
16.3 |
16.3 |
|
Water content (%) |
---------- |
6.7 |
6.7, 12 |
12 |
12 |
|
Confining pressure |
---------- |
---------- |
50, 100, 200 |
---------- |
|
|
Standard method |
ASTM D1557-12 |
ASTM D5333 |
ASTM D2850-87 |
ASTM D1883-99 |
|
Table 3 Collapse index results for biopolymer treated soil with various contents at a dry density of 1.36 /
|
Biopolymer concentration (%) |
Collapse index (%) |
|||||
|
Dry mix |
Wet mix |
|||||
|
Xanthan |
Sodium alginate |
Gelatin |
Xanthan |
Sodium alginate |
Gelatin |
|
|
0 |
13.48 |
13.48 |
13.48 |
13.48 |
13.48 |
13.48 |
|
0.5 |
8.00 |
9.00 |
10.30 |
7.14 |
7.22 |
9.50 |
|
1 |
6.10 |
6.72 |
8.20 |
4.19 |
5.77 |
7.60 |
|
1.5 |
4.14 |
5.00 |
6.45 |
3.05 |
4 |
5.59 |
|
2 |
3.57 |
4.09 |
4.86 |
2.15 |
3.04 |
4 |
|
3 |
1.85 |
2.73 |
3.96 |
1.24 |
2.14 |
2.73 |
|
4 |
1.27 |
2.05 |
2.90 |
0.3 |
0.95 |
2 |
Table 4 Collapse index results for xanthan gum, sodium alginate, and gelatin treated soil with various contents at a dry density of 1.63 kN /m3
|
Biopolymer content (%) |
Collapse potential (%) |
||
|
Xanthan |
Sodium alginate |
Gelatin |
|
|
0 |
9.27 |
9.27 |
9.27 |
|
0.5 |
5.00 |
5.84 |
6.73 |
|
1 |
3.95 |
4.40 |
5.4 |
|
1.5 |
2.45 |
3.37 |
3.74 |
|
2 |
2.11 |
2.65 |
3.05 |
|
3 |
1.05 |
1.50 |
2.72 |
|
4 |
0.3 |
0.50 |
1.71 |
Version 1
posted
You are reading this latest preprint version