Aim
To investigate the effects of different methods of breast milk expression on breastfeeding initiation, efficacy of breast milk extraction, and as a consequence, on the rate of exclusive breastfeeding at 6 months.
Design and setting of the study
In this open-label, randomized controlled trial, the participants were recruited at Xinhua Hospital affiliated to Shanghai Jiaotong University, certified as a Baby Friendly Hospital. This is a tertiary general hospital with a 75-bed obstetrics unit and approximately 3300-3500 deliveries annually. Being the Shanghai Critical Maternal Transfer Center, Shanghai Prenatal Diagnosis Center, and Shanghai Premature Birth Center, it is also a referral centre for high-risk pregnancies from other hospitals in Shanghai City and surrounding provinces.
Participants
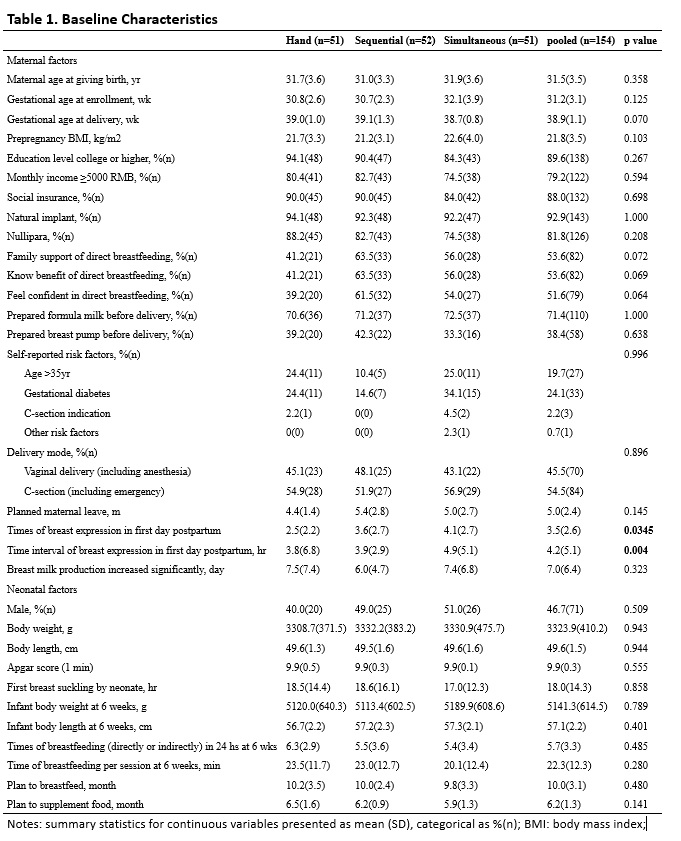
The participants were recruited from April 2018 to September 2019 through posters at an Obstetrics unit and in a delivery room of the hospital. Mother-neonate dyads were potentially eligible if the mother was aged 20-40 years, had attended antenatal education course in the hospital, committed to direct breastfeeding for at least 4 months in her maternity leave and were willing to express breast milk if required. The neonates had to have been delivered at gestational age ≥34 weeks and body weight ≥2000 grams with a 1-minute Apgar score ≥7. Dyads in which the woman had a history of smoking or regular alcohol drinking, or contraindications (such as human immunodeficiency virus or other infectious diseases) to direct breastfeeding, or the infant had congenital anomalies or was otherwise unsuitable for direct breastfeeding, were excluded. Ethical approval of this trial was obtained from the Institutional Review Board of Xinhua Hospital Ethics Committee Affiliated to Shanghai Jiaotong University School of Medicine on 25th Dec. 2017 (XHEC-C-2017-108-2). All participants provided written informed consent before any trial-related procedures, first during antenatal preparation classes during 30-37 weeks of gestation, and then if the mother-infant dyads matched the eligibility criteria, either just before delivery or within 2 hours after delivery, depending on the clinical situation of the baby. From recruitment onwards, the principles of good clinical practice were followed. This trial is registered with Chinese Clinical Trial Registry (http://www.chictr.org.cn), code ChiCTR1800018193. The reporting of the trial is guided by the extension to the CONSORT statement for multi-arm parallel-group randomized trials.33
Randomization and masking
Participants were randomly assigned into three groups (1:1:1) through a simple “lottery drawing” process without replacement. The information on group assignment, was pre-sealed in 155 opaque envelopes which were opened in the presence of two investigators. The three groups were defined according to the methods of breast milk expression in the absence of infant sucking, namely hand expression, sequential pumping (Avent SCF902/11 single electric breast pump, Philips Invest, China) and simultaneous pumping (Avent SCF303/01 bilateral electric breast pumps, Philips Invest, China) in the first 6 months after birth.
Hand expression means no vacuum sucking. The sequential Avent SCF902/11 single electric breast pump system provided five-gear vacuum pressure (5 to 1) labeled from -250 to -130 mmHg. The simultaneous pumping Avent SCF 303/01 system provided two mode of suction: Stimulation and Expression, both in four-gear mode. Vacuum pressure in Stimulation mode was from -150 to -80 mmHg, and from -250 to -170 mmHg in Expression mode. The suction frequency was 1.0-2.5 Hz in Stimulation mode and 0.71-1.05 Hz in Expression mode accordingly, while the single electric breast pump only provided constant vacuum pressure.
This was an open-label trial with blinded outcome assessment. Nurses collecting the data and questionnaire information, and statisticians conducting the analysis were blinded to group allocation.
Procedures
Recruitment and randomization occurred immediately (within 30 mins) after birth in the delivery/recovery room if a decision was made based that the neonates should be separated from their mothers for ≥6 hours. The common causes of mother and infant separation include preterm birth, neonatal asphyxia, maternal or neonatal suspicious infections (such as maternal fever before delivery, meconium-stained amniotic fluid) and complications of delivery. Physical examination and laboratory tests in pregnancy were carried out and demographic data were obtained.
All participants were trained in manual expression of breast milk during mother-baby separation according to guidelines for initiating milk supply in the absence of infant suction.34 Women were asked to begin breast milk expression at 2 hours after birth and continue with an interval of 2-3 hours between subsequent sessions (each comprising10 min of pumping per breast per session) until initiation of direct breastfeeding. After the infants were able to suck, the usage of breast pumping was restricted to remove excessive breast milk, to alleviate the fullness of breast milk, to donate breast milk, or to discard breast milk likely to be contaminated with medications. The number of sessions of breast pumping per day was expected to decrease gradually as lactation became established in healthy mother-infant dyads.
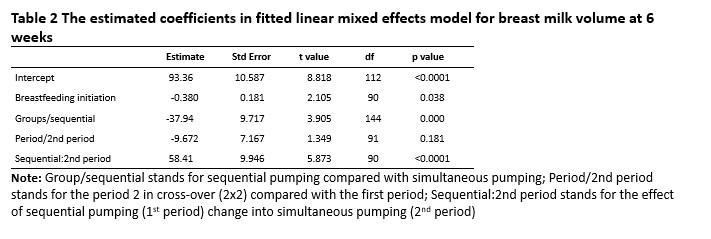
The 10-minute expression volume of breast milk per breast at 3 days and at 6 weeks postpartum were recorded, and the mother-infant dyads were followed up at 4 and 6 months after birth (Figure 1).
All women in this study stayed in the hospital for more than 48 hrs after delivery and were discharged at the 3rd day. On the 3rd day, the two breasts were sequentially or simultaneously pumped for 10 minutes per breast. This was repeated 2-3 hours later, using the same type of pumping (Figure 1).
At 6 weeks postpartum, the mothers were followed-up and asked to breastfeed directly first, and one hour later each breast was pumped for 10 minutes per breast with one of the two types of electric pump. After a clinical review, the mothers were asked to breastfeed their infants directly again, and one hour later had milk pumped with the other type of electric breast pump (crossed over trial).
The participants also joined an online trial member group through WeChat (Tencent, China), a mobile phone App that facilitates instant communication between participants and investigators. One investigator was assigned to answer questions from the trial group and provide professional advice. The information on breastfeeding at 4 and 6 months was collected through online questionnaire.
Outcomes and data collection
The two primary outcomes were (1) Time for lactation initiation: also known as time for breastfeeding initiation, defined as the time of the infant was provided successfully direct breastfeeding or with expressed breast milk between delivery and hospital discharge and (2) Volume or weight of expressed breast milk in 20 min (10 min per breast) at three days or six weeks postpartum. To assess the effects of manual breast expression, milk transfer was measured at 3 days and at 6 weeks postpartum. The infants were weighed just before and after breastfeeding to determine milk transfer to the infant in the obstetrics ward. This method had been validated and was used in lactogenesis research.9,35 Test weights of infants were obtained in duplicate using an electronic scale (accuracy=0.1 g). Each weight was recorded when the measurement was stable for 10 seconds. A density factor of 1.03 g/ml was used in conversion between weight and volume of breast milk.
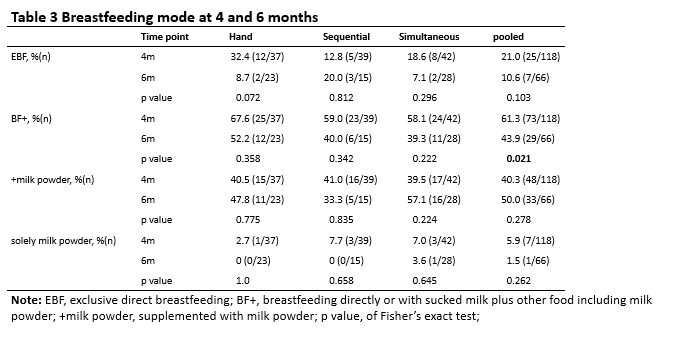
The secondary outcomes were the rates of exclusive breastfeeding at 4 and 6 months. The mode of feeding was divided into seven categories: direct breastfeeding, direct breastfeeding plus expressed breast milk, direct breastfeeding plus expressed breast milk and formula milk powder, direct breastfeeding plus formula milk powder, merely expressed breast milk, expressed breast milk plus formula milk powder, total formula milk powder. The data on self-reported mode of feeding were collected online.
The reported adverse events during breast pumping include those reported by mothers and from device. General adverse events are those, which patients or trial participants reported other than during breast pumping or were observed by clinicians.
Sample size and Statistical analysis
According to unpublished data from a pilot comparison between two groups of lactation-established women that each applied one of the two types of breast pumps, there was a difference in breast milk volume of around 15 ml with a standard deviation of 40 ml. To detect a difference of this magnitude, with a statistical power of 0.8 and significance level of 0.05, the sample size was estimated to be 90 participants in each group. Considering the limited number of potentially eligible participants, the target sample size was revised to 50 participants in each group, and a cross over design of breast pump types used at 6 weeks was incorporated in the trial design.
The data were analysed according to intention-to-treat (ITT). The continuous data were presented as mean±SD and the categorical data were presented as counts and percentages. To compare baseline characteristics, two-tailed one-way ANOVA was conducted for variables with a normal distribution and for other variables the Wilcoxon signed rank (Mann-Whitney U) test. For comparisons between the three trial groups, the nonparametric Kruskal-Wallis rank sum test and the chi-square test were applied for continuous and categorical data, respectively. Tukey honest significant differences were calculated in multiple comparisons adjusted with p values. The paired student’s t test was applied to compare the milk volumes between left and right breasts or between the two types of breast pump. The log-rank test was applied to compare the time required for breastfeeding initiation (event=1). Linear mixed-effect modelling (lme4 package) was used to analyze the association between the expressed milk volume and co-variates. p<0.05 was considered statistically significant. Statistical analyses were done using R (version 3.3.3).36