Background
Quality control of the herbal medicines (HMs) still hits technical bottlenecks due to their complicated multi-step pharmaceutical processes. Nuclear magnetic resonance (NMR) spectroscopy could serve as a powerful technique able to obtain comprehensive information of chemical compounds in the manufacturing process of HMs by a single analytical test. In this study, ethanol precipitation of Salvia miltiorrhiza Radix et Rhizoma (Danshen in China), a critical purification process of a particularly commonly used herbal medicinal material, was employed as the research subject.
Methods
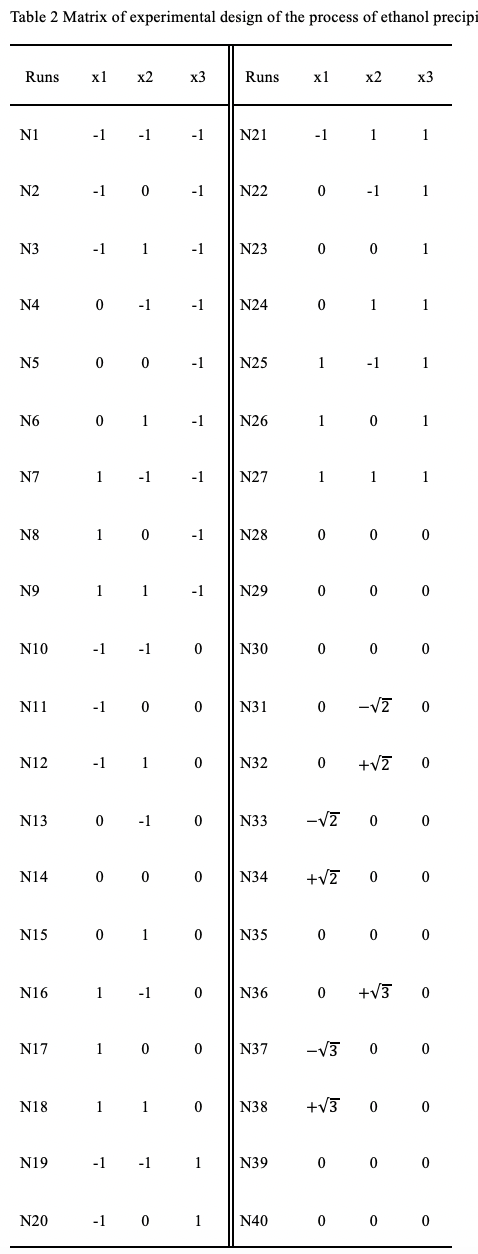
Processed with different variables, including ethanol-material ratio, apparent ethanol concentration and moisture concentration of the raw material, 40 lab-scale experiments of ethanol precipitation were orchestrated by a full factor design with additional central composite circumscribed experiments. After 1H NMR tests, chemometrics methods, including orthogonal partial least squares-discriminant analysis (OPLS-DA), principle component analysis (PCA) and multiple linear regression (MLR), were applied to analyze the spectra of samples.
Results
16 metabolites were recognized as the most critical potential chemical markers in the process of Danshen ethanol precipitation by 1H NMR spectra and OPLS-DA, including γ-aminobutyrate, pyroglutamate, succinate, acetate, malonate, fructose, sucrose, raffinose, stachyose, mannotriose, danshensu, lithospermic acid, protocatechuic aldehyde, rosmarinic acid, salvianolic acid A and B. PCA reasonably distinguished all the process intermediates as a function of different process variables adopted during the ethanol precipitation. MLR made the statistical interpretation by quadratic equations to characterize how do significant variables affect the chemical markers.
Conclusions
Our study indicates that NMR can track detailed potential chemical markers of ethanol precipitation process, which could promote the further understanding of ethanol precipitation process and open up possibilities to develop a more effective process quality control and consistency evaluation methodology of an HM, through providing a more in-depth and improved theoretical process understanding and characterization.