The aim of this study is to investigate the impact of histology in the efficacy of systemic treatment in a real-world database MPM. Our results show that epithelioid tumors presented better outcomes when received chemotherapy, irrespective of the platinum agent.
Histology has been broadly studied in MPM and well recognized as a prognostic factor [19, 20]. The two prognostic scoring systems classically used in mesothelioma were developed previous to the routine use of pemetrexed and both scores included nonepithelial histology as predictor of poor survival (EORTC and CALGB) with a difference in median survival around 3 months between epithelioid and nonepithelioid. However, the role of histology as predictive factor of efficacy to treatment with chemotherapy in advanced MPM has not been well address in the clinical trials. In the era of pemetrexed treatment, only a retrospective analysis of 131 patients treated with chemotherapy demonstrated that epithelioid histology was assessed with clinical benefit from first line chemotherapy [21].
Recently, the study CheckMate 743 comparing chemotherapy versus nivolumab plus ipilimumab in untreated patients demonstrated that the combination of immunotherapy was superior in terms of survival [15]. In the trial patients were stratified by histology (epithelioid versus nonepithelioid) including 75% of patients with epithelioid histology. In a preplanned subanalysis, the improvement of immunotherapy over chemotherapy was clearly superior in nonepithelioid patients with a median OS of 18.1 months with immunotherapy and 8.8 months with chemotherapy. For the overall population no differences in terms of PFS were detected. Authors concluded that survival benefit with nivolumab plus ipilimumab over chemotherapy was observed regardless of histology with better outcomes for chemotherapy in epithelioid histology. The presence of PD-L1 also predicted improvement with nivolumab plus ipilimumab over chemotherapy, but according to the authors of the study, PD-L1 results were descriptive only, precluding definitive conclusions.
The pivotal trial EMPHACIS published by Vogelzang demonstrated that combination of therapy with cisplatin plus pemetrexed was superior to cisplatin alone [3]. In the trial 68% of patients were epithelioid histology, but no data about the efficacy by histological subgroups were reported. Similarly, in a subsequent phase IV trial evaluating the role of pemetrexed in mesothelioma, the efficacy by histology was not reported [22]. Raltitrexed, the other antimetabolite approved in malignant mesothelioma also demonstrated improved in survival in combination with cisplatin and about two thirds of patients were epithelioid [23]. In this trial patients with nonepithelial tumors presented worse prognostic in univariate and multivariable analysis, but no data were published regarding the predictive role of the treatment with histology. Carboplatin in association with pemetrexed is an alternative option for patients who may not tolerate cisplatin [6, 7]. In the larger of two randomized phase II studies response was observed in patients with epithelial or mixed histology, but no response was registered in patients with sarcomatoid mesothelioma.
In order to improve the outcomes of the chemotherapy, the addition of antiangiogenics or TTFields has been explored. The MAPS trial demonstrated that survival was significantly extended with the addition of bevacizumab to chemotherapy [4]. In a preplanned subgroup analyses, the effect on survival of the bevacizumab containing regimen compared with standard chemotherapy was homogeneous when the analysis was stratified by histology subtype, moreover, the effect was more pronounced in patients with sarcomatoid or mixed histology (HR for OS of 0.82 (0.64–1.06) for epithelioid and 0.64 (0.40–1.02) for sarcomatoid). However, another two trials with antiangiogenic have failed in demonstrate benefit in patients with mesothelioma. The phase II LUME/Meso designed to assess the efficacy of nintedanib plus chemotherapy, demonstrated evident benefit in epithelioid histology, but not in biphasic, however the number of patients with biphasic histology was too low to provide a reliable estimate of the treatment effect [24]. Also, the addition of cediranib to chemotherapy improved PFS and there was no difference in the effect of treatment by histological subtypes [25]. In the STELLAR trial TTFields delivery system in combination with chemotherapy for first line leads to a median OS and PFS longer than historical control [5]. In a post-hoc analysis, OS and PFS were longer in patients with epithelioid histology than in patients with other subtypes (OS 21.2 vs 12.1 months and PFS 8.3 vs 6.5 months, respectively).
Less evidence of the difference in the efficacy of treatment according to histology has been evaluated in studies of previously treated patients. Vinorelbine has shown clinical activity in a phase II study and responses were observed in all three histologic subtypes of mesothelioma, including those with sarcomatoid and biphasic tumors [26]. More recently, lurbinectedin demonstrated no significant differences in PFS and OS concerning the impact of histology, suggesting that lurbinectedin is likely to equalize the prognosis of the mesothelioma subtypes [27].
The impact of immunotherapy in mesothelioma has been recently demonstrated. Initial studies with monotherapy suggested efficacy, but randomized trials in previously treated patients are controversial. However, in first line setting, a recent study demonstrated better outcomes for immunotherapy over chemotherapy. In these studies of immunotherapy, predictive factors of response have been more studied. Keynote 028 phase I trial, enrolled previously treated PD-L1 positive mesothelioma patients and showed 40% of clinical benefit for more than 6 months [10]. In the trial 72% of patients were epithelioid but no results according to histology subtypes were reported. In the INITIATE trial, a single arm phase 2 trial of nivolumab plus ipilimumab, disease control rate at 12 weeks was achieved by 68% [8]. The study included 86% of patients with epithelioid subtype and the small number of tumors with nonepithelioid histology did not allow a meaningful comparison between histological subtypes. MAPS2 trial also evaluated the addition of ipilimumab to nivolumab and demonstrated better outcomes for the combination [14]. Patients were stratified by histology with 85% of patients being epithelioid and responses were reported in all histological groups. PROMISE-meso failed in demonstrate superiority of pembrolizumab over chemotherapy in relapsed mesothelioma [11]. In this trial patients with nonepithelioid tumors had a non-significant poorer PFS and OS for pembrolizumab as compared to epithelioid. Two studies testing the combination of chemotherapy plus durvalumab have been reported. The Australian DREAM trial reported a 6 months PFS of 31% [28]. In the trial, 83% of patients were epithelioid and in a post-hoc analysis, responses were observed in all histological subtypes. The US PrE0505 trial also reported median OS of 20.4 months and this trial included 74.5% of patients with epithelioid tumors, but no data about the efficacy of the treatment by histology were reported [29].
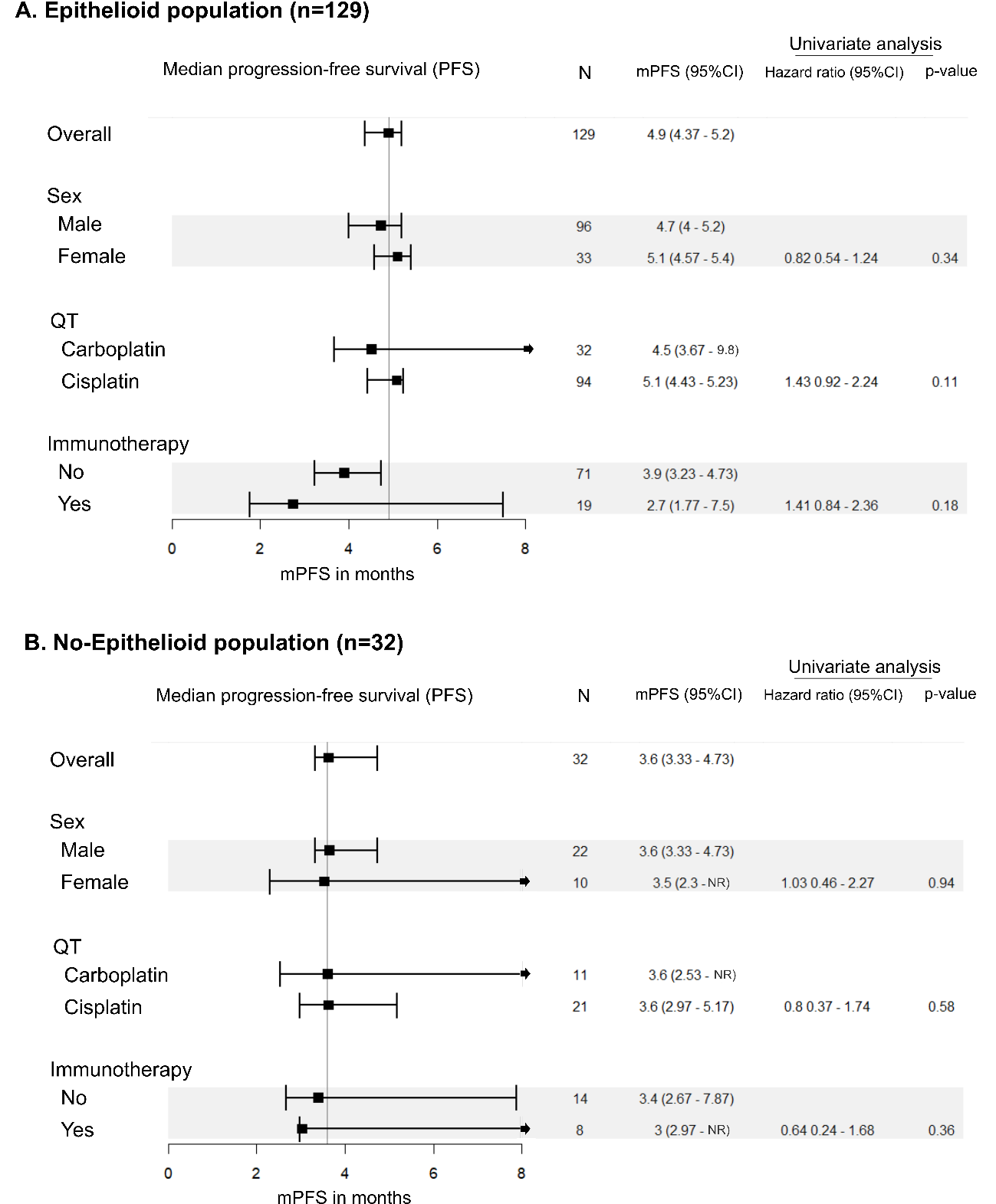
Since the publication of the CheckMate 743 pointing differences in efficacy of chemotherapy according to histology, we sought to perform a retrospective analysis of the efficacy of the chemotherapy with histology at our institution. We evaluated 189 patients and we found, in agreement with other series that histology is a strong prognostic factor with a difference in median OS of 11.7 months (21.3 months in epithelioid versus 9.6 months in nonepithelioid). In our real-world series, we could demonstrate that histology is a prognostic factor for PFS in favor of epithelioid histology in patients treated with chemotherapy with a median PFS of 4.8 versus 3.6 months (p = 0.03). We detected that patients with epithelioid histology treated with cisplatin had higher benefit than patients treated with carboplatin (4.5 months versus 3.6 months for patients treated with carboplatin). Despite the numerically higher PFS in patients with epithelioid tumors treated with cisplatin, there was no significant interaction between platinum agent and histology in Cox models, suggesting that histology is not a determinant of platinum agent sensitivity.
At the moment, the trials that led approval of pemetrexed and raltitrexed in malignant mesothelioma did not reported difference in efficacy of the chemotherapy according to histology, and the addition of bevacizumab demonstrated efficacy of treatment in all subgroups with better outcomes for sarcomatoid and biphasic tumors. Only the combination of carboplatin plus pemetrexed reported no responses in sarcomatoid mesothelioma. Our results, in accordance with previous studies confirms that histology is a prognostic factor. In our series epithelioid histology was a significant determinant of PFS in patients treated with chemotherapy, confirming one of the conclusions of the CheckMate 743 pointing worse efficacy of chemotherapy in nonepithelioid patients. In our study we included a small cohort of patients treated with immunotherapy (27 patients) and in this cohort we did not detect differences in PFS according to histology.
Our results have some limitations. This is a real-world series including all patients treated at one single institution. In our series, the number of epithelioid histology patients was high (76%) but this percentage is in line with the proportion of epithelioid patients included in clinical trials. We compare the impact of the treatment by histology but the number of patients with no epithelioid included is small.
In conclusion, in our series we confirm that histology is a prognostic factor and patients with nonepithelioid tumors had worse survival. Patients with epithelioid histology presented better PFS than patients with nonepithelioid tumors. Ongoing studies combining checkpoint inhibitors plus chemotherapy are evaluating the impact of histology in the outcomes.